The Rise of Patient Advocacy
Patient advocacy in the United States has evolved from a grassroots movement to a strategic partner in drug development. Early milestones include the actions of ACT UP and the Treatment Action Group, which helped create the FDA’s Accelerated Approval pathway, allowing life‑saving drugs for serious diseases to be approved on surrogate endpoints. The 1983 Orphan Drug Act, championed by rare‑disease advocates, introduced tax credits, grant funding, and seven‑year market exclusivity, spurring investment in treatments for uncommon cancers. More recently, the FDA’s Patient‑Focused Drug Development (PFDD) program institutionalizes patient input, shaping eligibility criteria, endpoints, and trial logistics. Advocacy groups now co‑fund early‑stage research, co‑design protocols, and embed patient‑reported outcomes (PROs) and decentralized trial designs, which improve enrollment and retention. Initiatives such as the Alliance for Clinical Trials in Oncology and the AACR Scientist↔Survivor Program demonstrate how survivor voices influence protocol review, recruitment strategies, and the inclusion of under‑represented populations, making modern oncology trials more patient‑centered and efficient.
Building Partnerships: Clinical Research Alliances
 The Clinical Research Alliance (CRA) is an academic clinical‑research organization anchored at the UNC School of Medicine that coordinates multi‑site oncology trials across a nationwide network of investigators and cancer. Its structure pairs a central coordinating hub with regional sites, allowing a single point of contact for protocol development, regulatory compliance, data management, budgeting, and contract negotiation. By leveraging established alliances with sponsors, vendors, and community hospitals, CRA streamlines trial start‑up and accelerates enrollment, especially for complex cancer studies that require diverse patient populations.
In multi‑site oncology trials, CRA provides standardized operating procedures, real‑time data monitoring, and adaptive design expertise that reduce variability between sites and shorten timelines. The alliance’s centralized biostatistics and FDA strategy teams ensure that trial modifications, such as biomarker‑guided adaptations or real‑world evidence arms, are implemented without compromising scientific integrity. This model supports rapid translation of innovative therapies—like those emerging from Hirschfeld Oncology—into practice.
Collaboration with patient‑advocacy groups is integral to CRA’s mission. Advocacy organizations co‑design eligibility criteria, contribute patient‑reported outcome measures, and help create plain‑language study summaries. Through advocacy‑driven navigation programs and decentralized trial options, CRA improves enrollment of under‑represented populations, enhances retention, and aligns trial endpoints with what matters most to patients.
Recruiting the Right Participants
 Clinical trial recruitment software leverages artificial‑intelligence and real‑time electronic health‑record integration to automatically screen thousands of patients against study eligibility criteria, dramatically cutting the time needed to identify suitable candidates. By providing instant, bias‑free matching and centralized dashboards, the platform helps oncology sites such as Hirschfeld Oncology meet enrollment targets, maintain trial accuracy, and reduce costly delays. Integrated omni‑channel outreach tools engage patients across web, mobile, and provider networks, improving diversity and allowing rapid communication of trial opportunities.
To increase enrollment, raise physician and patient awareness through a dedicated, easy‑to‑navigate trial website that lists eligibility, expectations, and searchable site locations. Build a centralized patient database that captures individuals screened out of one study and automatically flags them for other appropriate trials, enabling cross‑trial recruitment without extra spend. Implement a real‑time enrollment‑management platform that streamlines site‑to‑sponsor communication, tracks slot availability, and speeds up patient allocation. Design trials with patient‑centric protocols—offering remote visits, travel assistance, and flexible scheduling—to reduce logistical barriers and improve participation.
Patient recruitment companies specialize in finding and enrolling eligible patients by leveraging omni‑channel outreach, electronic health‑record mining, and advocacy networks. Leading firms such as IQVIA Patient Recruitment, M&B Sciences (Clara Health), Antidote, AutoCruitment, and Deep 6 AI accelerate enrollment, lessen site burden, and improve diversity—crucial for oncology trials such as those for pancreatic cancer. Partnering with these specialists allows Hirschfeld Oncology to secure timely enrollment while maintaining regulatory compliance and a patient‑centric experience.
Recruitment and retention challenges include low awareness, stringent criteria, logistical burdens, and safety concerns. Solutions involve culturally sensitive communication strategies, community patient registries, flexible scheduling or monitoring, financial assistance, transportation support, and regular progress updates. A dedicated recruitment team that collaborates with investigators, nurses, and patient‑advocacy groups ensures continuous engagement from enrollment through study completion.
The Advocate’s Voice in Trial Design
 Patient advocacy has reshaped oncology trial design by putting the lived experience of cancer patients at the table.
Patient‑reported outcomes (PROs) have moved from optional add‑ons to core endpoints in many Phase III studies. Guided by the FDA’s Patient‑Focused Drug Development guidance, advocates have helped embed instruments such as PRO‑CTCAE and FACT‑G, allowing symptom burden and quality‑of‑life to be measured alongside tumor response. This shift improves relevance for patients and regulators alike.
Eligibility and endpoint refinement is another arena where advocates make a measurable impact. By reviewing protocols early, groups like the AACR Scientist↔Survivor Program® and the Alliance for Clinical Trials in Oncology have broadened eligibility—e.g., allowing early‑stage pancreatic cancer patients into immunotherapy trials and expanding age limits for rare‑disease studies. Their input also drives the inclusion of meaningful endpoints such as patient‑reported outcome measures (PROMs) and molecular residual disease (MRD) markers, which better reflect what patients value.
Advocate compensation and roles are evolving from volunteerism to professional engagement. Independent patient advocates typically charge $100‑$500 per hour, with a common range of $200‑$300; the midpoint, $250 per hour, serves as a budgeting benchmark. Many advocates are compensated through bundled service agreements or retainers, and Medicare may cover navigation services, reducing out‑of‑pocket costs for patients.
Key Q&A
- What is a cancer patient advocate? A cancer patient advocate—also called a patient navigator—is a trained professional who assists individuals and families navigate the complex cancer care system, coordinates appointments, provides education, and serves as a bridge between patients and researchers.
- What is the average cost of a patient advocate? Independent advocates charge $100‑$500 per hour; the midpoint is about $250 per hour.
- What are the 4 methods of randomization? Simple randomization, block randomization, stratified randomization, and covariate‑adaptive randomization (minimization).
- 4 phases of clinical trials Phase I (safety/dosing), Phase II (efficacy), Phase III (comparison to standard care), Phase IV (post‑approval surveillance).
- Do cancer clinical trials use placebos? Rarely; most use active controls, and placebo arms are employed only under strict ethical justification.
- How to find a cancer advocate? Call the American Cancer Society helpline (1‑800‑227‑2345), use the ACS CARES™ app, ask your oncologist or hospital patient‑services office, or join disease‑specific advocacy groups such as PanCAN.
- What assistance is available for cancer patients? Financial aid from government programs, charitable foundations, and nonprofit organizations cover medicines, transportation, and other costs; advocates help patients navigate these resources.
By integrating patient voices early, ensuring fair compensation for advocates, and aligning trial design with real‑world priorities, oncology research becomes more efficient, inclusive, and patient‑centered.
Accelerated Approvals, Adaptive Designs, and Real‑World Evidence
 Patient advocacy groups such as ACT UP and the Treatment Action Group were instrumental in establishing the FDA’s Accelerated Approval pathway, which permits drugs for serious diseases to be approved on surrogate endpoints such as objective response rate or biomarker evidence. This pathway, first championed by groups like ACT UP and the Treatment Action Group, mandates post‑approval confirmatory studies to verify long‑term safety and efficacy, ensuring rapid patient access while preserving regulatory oversight.
Adaptive trial designs—including platform, basket, and multi‑arm multi‑stage (MAMS) trials—have become standard in oncology. Platform trials (e.g., STAMPEDE, I‑SPY‑2) allow simultaneous testing of multiple agents and dynamic modification of arms based on interim data. Basket trials enroll patients across tumor types sharing a common molecular target, expanding options for rare or orphan cancers. MAMS designs enable early stopping for futility and seamless phase transitions, reducing patient numbers and development timelines. Patient advocates have driven the inclusion of patient‑reported outcomes and flexible eligibility criteria within these adaptive frameworks.
Real‑world evidence (RWE) now complements traditional trial data. Electronic health records, claims, and cancer registries supply external control arms and post‑market safety monitoring, accelerating approvals and reducing reliance on placebo groups. Advocacy groups such as the Pancreatic Cancer Action Network and AACR’s Scientist↔Survivor Program® champion RWE integration to ensure that trial findings reflect everyday clinical practice.
Phase II clinical trial design – Phase II trials enroll 100–300 patients to assess efficacy signals (e.g., objective response rate, progression‑free survival) and safety, often using randomized or Simon two‑stage designs with predefined stopping rules.
Clinical trial recruitment software – AI‑driven platforms screen EHRs in real time, match patients to eligibility criteria, and deliver multi‑channel outreach, boosting enrollment speed, diversity, and trial efficiency for sites like Hirschfeld Oncology.
Accelerated approvals and adaptive designs – Advocacy‑driven accelerated approvals, combined with adaptive platform, basket, and response‑adapted trials, enable faster drug access while post‑market RWE confirms benefit.
Recruitment and retention challenges and solutions – Low awareness, strict criteria, and logistical burdens hinder enrollment. Solutions include culturally sensitive communication, patient registries, decentralized visits, financial assistance, and continuous engagement through advocacy‑partnered recruitment teams.
Digital & Decentralized Trials
 Virtual Patient Advocate A virtual patient advocate is a trained healthcare professional who provides personalized support remotely through secure video calls, phone, or digital platforms. For pancreatic cancer patients at Hirschfeld Oncology, the VPA helps translate complex treatment options, coordinate appointments, and ensure clear communication between the medical team and the patient’s family. By leveraging AI‑enhanced tools and telehealth integration, the advocate can track medication schedules, flag missed labs, and assist with insurance or billing questions in real time. This digital liaison also connects patients with clinical trial information, financial assistance programs, and supportive services, reducing the stress of navigating a multidisciplinary care plan. Ultimately, a virtual patient advocate empowers patients to make informed decisions and stay engaged throughout their cancer journey, regardless of geographic location.
Patient Advocacy Conferences Patient advocacy conferences such as the AACR Annual Meeting and the ASCO Annual Meeting provide vital platforms where patients, caregivers, and advocates can engage directly with leading researchers and clinicians. At the AACR Annual Meeting, dedicated sessions like the Scientist↔Survivor Program® and the Advocacy Partners Pavilion allow advocates to share lived‑experience insights that shape research priorities and clinical practice. The ASCO‑Sponsored Patient Advocacy Booth offers nonprofit organizations a space to showcase resources, connect with the oncology community, and distribute educational materials. Hirschfeld Oncology encourages its pancreatic‑cancer patients and families to attend these events, offering guidance on registration, travel, and how to maximize networking opportunities. By participating, patients gain access to the latest scientific advances, supportive services, and a collective voice that drives more compassionate, patient‑centered cancer care.
Phase III Clinical Trial A Phase III clinical trial is a large, multi‑center study that evaluates the safety and efficacy of a new treatment compared with the current standard of care. It typically enrolls several hundred to several thousand patients who have the disease of interest and can last from one to four years. The trial is usually randomized and blinded, allowing researchers to assess outcomes such as overall survival, disease‑free survival, and side‑effect profiles with statistical rigor. Data generated from Phase III trials form the bulk of the evidence required for FDA approval and labeling of a new drug. Successful Phase III results are essential before a therapy can become widely available to patients, including those treated at specialized centers like Hirschfeld Oncology.
Stage 4 Cancer Clinical Trials Stage 4 (metastatic) cancer clinical trials evaluate new drugs, combinations, or treatment strategies in patients whose disease has spread beyond its original site. These studies often focus on targeted therapies, immunotherapies, or novel dosing regimens—such as regorafenib, fruquintinib, or pembrolizumab‑based combos—to improve survival and quality of life while monitoring safety. Participation can give patients access to cutting‑edge treatments that are not yet widely available, as illustrated by the dramatic response of a stage 4 lung‑cancer patient to an immunotherapy‑radiation trial. At Hirschfeld Oncology, we actively enroll eligible pancreatic‑cancer patients in multicenter trials that incorporate the latest scientific advances and personalized medicine. Our multidisciplinary team guides you through eligibility, consent, and supportive care so you can explore every promising option while maintaining comfort and dignity.
Wearable & Mobile Health Technologies & Wearable devices and smartphone apps now feed continuous physiologic data and ePROs into trial databases, reducing site visits and patient burden. These tools enable real‑time monitoring of symptom burden, activity levels, and treatment side effects, enhancing data richness for adaptive trial designs.
Decentralized Trial Models Decentralized and hybrid trial designs combine remote consent, telemedicine visits, and home‑based drug administration, expanding access for patients in rural or underserved areas. By partnering with patient‑advocacy groups, sponsors can identify community sites, provide navigation services, and ensure equitable enrollment across diverse populations.
Advocacy for Diversity, Inclusion, and Policy Change
 [Patient advocacy groups] are now central to improving the enrollment of under‑represented populations in oncology trials. [The FDA’s Diversity Action Plans], sharpened by voices from organizations such as the Alliance for Clinical Trials in Oncology and the American Cancer Society Cancer Action Network (ACS CAN), require sponsors to develop recruitment strategies that specifically target racial, ethnic, and geographic minorities. By leveraging community‑based outreach, culturally‑tailored consent materials, and [Patient navigation programs] that address transportation and financial barriers, trials have increased enrollment of Black, Hispanic, and rural patients, producing data that are more generalizable to real‑world practice.
Legislative advocacy and funding have been amplified through coordinated efforts of ACS CAN and other coalitions. ACS CAN, the non‑partisan advocacy arm of the American Cancer Society, lobbies federal and state policymakers for increased NIH and NCI research budgets, supports the Clinical Trial Modernization Act, and promotes policies that reduce patient‑out‑of‑pocket costs. These policy wins translate into tangible resources for trial sites, including grant funding for patient‑navigation services and tax credits that incentivize inclusive trial designs.
Answers to key questions
- ACS Cancer Action Network: Founded in 2001 and based in Washington, DC, ACS CAN mobilizes volunteers to lobby for stronger cancer‑research funding, broader care access, tobacco‑control measures, and legislation such as the Clinical Trial Modernization Act, thereby shaping a health‑care environment that supports cutting‑edge treatments like those offered at Hirschfeld Oncology.
- What help is available for cancer patients? Patients can draw on state‑based benefits, disability payments, and unemployment support, as well as emergency grants, low‑interest loans, and hardship funds from charities and patient‑advocacy groups. Financial counselors and cancer‑support helplines assist with navigating these resources.
- How to find a cancer advocate? Call the ACS 24‑hour helpline (1‑800‑227‑2345) or use the ACS CARES™ app for a matching volunteer; ask oncologists or Hirschfeld Oncology’s patient‑services office for a navigator referral; explore Hope Lodge® and disease‑specific groups like PanCAN for mentorship.
- Which is better, CRC or CRA? CRAs earn higher salaries, travel to multiple sites, and focus on monitoring compliance, while CRCs work on‑site, manage enrollment, and interact directly with patients. The better choice depends on personal career goals and preferred work style.
- Cancer advocate insurance: This supplemental plan pairs health coverage with proactive genetic testing, counseling, and a dedicated oncology nurse advocate who assists with treatment navigation, clinical‑trial enrollment, and recurrence monitoring, offering cash benefits and services to offset out‑of‑pocket expenses.
Through these coordinated advocacy actions—targeted recruitment, mandated diversity plans, and strong legislative support—clinical trials are becoming more inclusive, efficient, and aligned with patient priorities.
Putting It All Together at Hirschfeld Oncology
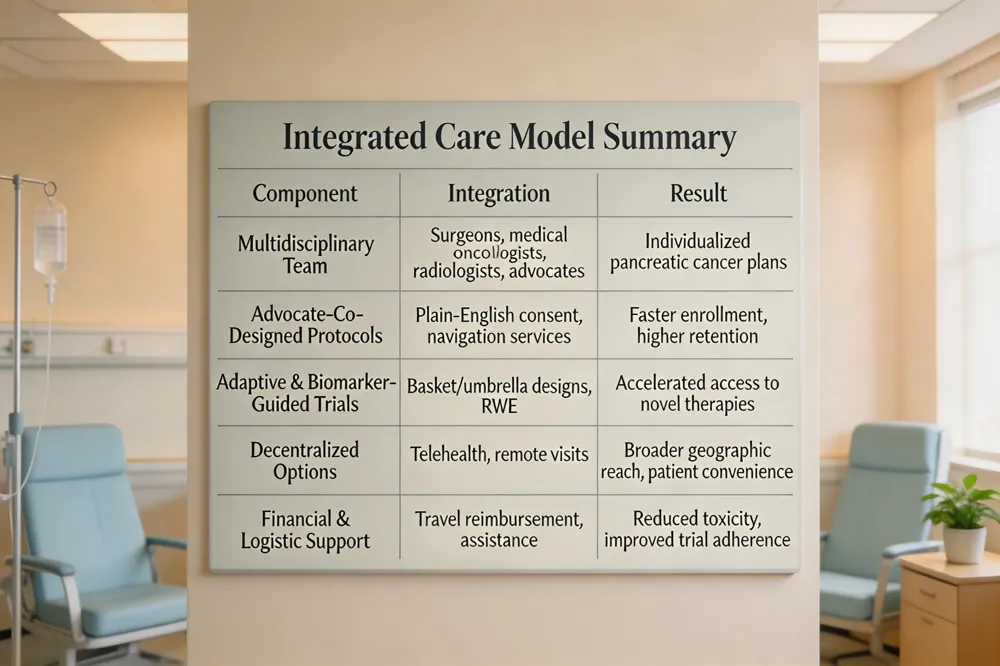 Hirschfeld Oncology’s multidisciplinary care model brings surgeons, medical oncologists, radiologists, and dedicated patient‑advocacy staff together to create individualized pancreatic cancer plans. Patient advocates co‑design trial protocols, translate consent language into plain‑English, and run navigation services that offset travel, financial toxicity, and scheduling barriers—an approach echoed by the Alliance for Clinical Trials in Oncology, AACR’s Scientist↔Survivor Program®, and NCI‑mandated Patient‑Focused Drug Development meetings. This integration accelerates enrollment, improves retention, and ensures emerging therapies mirror real‑world patient priorities.
How long do cancer clinical trials last? Phase I studies often finish within months, Phase II may run up to two years, Phase III can extend four to five years or more, and Phase IV post‑approval studies may continue for several years; individual participants typically complete their involvement sooner as enrollment is staggered.
What is the most common clinical trial design? The randomized, parallel‑arm controlled trial—often double‑blind—remains the gold standard, balancing known and unknown confounders across arms while each participant stays on a single treatment throughout the study.
What assistance is available for cancer patients? Federal and state programs, charitable foundations, and nonprofit groups (e.g., Patient Advocate Foundation, PAN Foundation, ACS CAN) offer financial counseling, travel reimbursement, co‑pay assistance, and emergency grants. Patient‑advocacy teams help patients navigate these resources, apply for hardship funds, and connect with community‑based support.
Future outlook for pancreatic cancer research Advocacy‑driven adaptive trial designs, biomarker‑guided basket and umbrella studies, and decentralized trial models are expanding access to novel immunotherapies and targeted agents. Real‑world evidence initiatives and patient‑reported outcome measures will further align drug development with the lived experiences of pancreatic cancer patients, shortening the path from discovery to approval.
A New Era of Patient‑Centered Oncology
Patient advocacy has reshaped oncology research, turning patients from passive subjects into active partners. Groups such as ACT UP, the Treatment Action Group, and the AACR Scientist↔Survivor Program have helped create accelerated‑approval pathways, co‑author protocols, and embed patient‑reported outcomes in phase‑III studies, boosting enrollment by 20‑30 % and expanding eligibility for under‑represented populations. Looking ahead, trial designs will increasingly use adaptive, biomarker‑driven master protocols, decentralized digital platforms, and real‑world evidence, all guided by patient input on endpoints, consent language, and logistical burdens. Researchers, clinicians, and sponsors are urged to formalize patient advisory boards, fund training programs, and share results. Patients are encouraged to join advocacy networks, provide feedback, and champion trial access.





.png)


.png)
.png)




