Setting the Stage for a New Era in Pancreatic Cancer Treatment
Pancreatic ductal adenocarcinoma (PDAC) has long been a therapeutic nightmare: over 80% of patients present with advanced disease, median overall survival after standard chemotherapy hovers around 11–12 months, and five‑year survival remains below 10%. The tumor’s dense desmoplastic stroma, pervasive KRAS mutations, and immunosuppressive microenvironment have limited the impact of single‑agent therapies. Hirschfeld Oncology’s multidisciplinary model confronts these hurdles by integrating surgical, medical, radiation, and supportive‑care specialists, while simultaneously deploying rapid molecular profiling to match patients with biomarker‑driven trials. This collaborative infrastructure enables seamless enrollment in complex regimens that pair cytotoxic backbones (FOLFIRINOX or gemcitabine‑nab‑paclitaxel) with targeted agents (KRAS G12C/D inhibitors, PARP inhibitors, HER2‑ADCs), stromal modifiers, or immune‑checkpoint blockers. Such combination strategies are now essential because they attack multiple resistance pathways—upstream, downstream, and orthogonal to KRAS—while leveraging chemotherapy‑induced DNA damage and immune activation, offering the best chance to extend survival and improve quality of life for PDAC patients.
Foundations of Integrated Treatment
 The most successful treatment for pancreatic cancer is a multimodal approach that blends surgery, systemic chemotherapy, and, when appropriate, radiation therapy. For resectable disease, curative‑intention pancreaticoduodenectomy (Whipple) or distal pancreatectomy is performed, followed by adjuvant multi‑agent chemotherapy such as FOLFIRINOX or gemcitabine plus nab‑paclitaxel, which improve median overall survival to 11–12 months. In borderline‑resectable or locally advanced cases, neoadjuvant chemotherapy (the same regimens) is given first to shrink tumors and increase the likelihood of an R0 resection; stereotactic body radiation therapy or concurrent chemoradiation is added for local control, especially when margins are positive. Radiation is reserved for selected patients where surgical clearance is incomplete. While pancreatic cancer remains highly lethal, emerging targeted and immunologic agents—KRAS inhibitors, PARP inhibitors for BRCA‑mutated tumors, and checkpoint‑blockade combinations—have produced partial and, in rare instances, complete responses. Real‑world data and early‑phase trials now show that up to 5 % of patients achieve durable disease control, with several documented long‑term remissions after personalized combination therapy at multidisciplinary centers such as Hirschfeld Oncology.
Targeted KRAS‑Driven Therapies and Triple‑Regimen Breakthroughs
 KRAS mutation landscape in PDAC More than 90 % of pancreatic ductal adenocarcinoma (PDAC) tumors carry KRAS alterations; KRAS G12C is present in ~1–2 % while KRAS G12D accounts for 30–40 % of cases, providing a strong rationale for KRAS‑directed drugs.
FDA‑approved KRAS G12C inhibitors Sotorasib and adagrasib (both FDA‑approved) achieve objective response rates of 30‑40 % in early trials and are now being combined with gemcitabine‑based regimens to improve disease‑control rates.
Emerging KRAS G12D inhibitors MRTX1133 and the multi‑selective RAS‑ON inhibitor RMC‑6236 (daraxonrasib) show promising activity in pre‑clinical models and early‑phase studies, especially when paired with standard chemotherapy.
Triple‑targeted pre‑clinical regimen RMC‑6236 + afatinib + SD36 simultaneously blocks KRAS, EGFR family, and STAT3 pathways. Mouse orthotopic and PDX models displayed complete, durable tumor regression with no relapse for >200 days and minimal toxicity.
2026 FDA approvals and late‑stage trials Daraxonrasib is advancing in three global Phase III trials (RASolute 302‑304), including a post‑surgical adjuvant study. Novel stromal‑modifying agents and bispecific antibodies are also entering late‑stage evaluation.
Daraxonrasib pancreatic cancer Daraxonrasib (RMC‑6236) is an oral RAS‑ON inhibitor targeting G12X, G13X and Q61X mutants. Phase II data show a 35 % objective response, >90 % disease control, and median PFS ≈8.5 months with a favorable safety profile.
Pancreatic cancer triple therapy The RMC‑6236 + afatinib + SD36 trio achieves complete, lasting regression in pre‑clinical PDAC models, while a PD‑1 + gemcitabine/nab‑paclitaxel + stromal‑modifying regimen improves immune infiltration and response rates.
New pancreatic cancer treatment 2026 Late‑stage trials now test KRAS G12D inhibitors (e.g., INCB161734) with FOLFIRINOX, claudin‑18.2 bispecifics, and multi‑antigen T‑cell therapies, all showing ≥30 % response rates and durable disease control.
New pancreatic cancer treatment 2025 2025 saw the debut of CEA/PD‑L1 bispecific antibodies, the Phase III RASOLUTE 302 trial of daraxonrasib, and AI‑discovered STAT3 linker‑domain inhibitor striatal B, which synergizes with chemotherapy in pre‑clinical models.
PNAS pancreatic cancer 2026 PNAS reported that the daraxonrasib + afatinib + SD36 triple regimen produced complete tumor regression without resistance for >200 days and that the NQO1‑activated prodrug C29h selectively kills NRF2‑high PDAC, synergizing with gemcitabine and enhancing CD8⁺ T‑cell activity.
Immunotherapy, Vaccines, and Cutting‑Edge Clinical Trials
 Checkpoint‑inhibitor plus chemotherapy combos – Trials such as pembrolizumab or nivolumab paired with gemcitabine‑nab‑paclitaxel or FOLFIRINOX are underway to breach the dense stroma and improve progression‑free survival. Early phase II data show disease‑control rates of 45‑50% in microsatellite‑stable disease.
Personalized mRNA and neoantigen vaccines – Autogene cevumeran, an individualized mRNA vaccine encoding up to 20 tumor‑specific neoantigens, is being given after surgery with mFOLFIRINOX, eliciting immune responses in half of treated patients and extending recurrence‑free survival.
Adoptive cell‑therapy and CAR‑T approaches – Engineered T‑cell receptors targeting cryptic pancreatic peptides and CAR‑T cells against mesothelin or claudin‑18.2 are in phase I/II studies, showing tumor shrinkage in 20‑30% of participants.
Key trial sites – Hirschfeld Oncology , Mayo Clinic, MD Anderson and the NCI coordinate multi‑institutional protocols, offering biomarker‑driven combos, stromal‑modulating agents and immunotherapy.
Answering common queries: Current immunotherapy trials combine checkpoint inhibitors with chemotherapy or vascular‑activating agents to turn “cold” tumors “hot.” The most promising Phase III studies include POLO (olaparib for BRCA‑mutated) and NAPOLI‑3 (liposomal irinotecan). Patients can locate nearby trials via ClinicalTrials.gov or NCI Trial Finder; Mayo and MD Anderson list active studies on their websites.
Combination Strategies for Advanced and Metastatic Disease
 Intensified Chemotherapy Backbones FOLFIRINOX and gemcitabine‑nab‑paclitaxel remain NCCN‑recommended first‑line regimens for metastatic PDAC, extending median OS to ~11–12 months. Sequential maintenance (e.g., PARP inhibition in BRCA/PALB2‑mutated disease) is increasingly used to prolong response while limiting toxicity.
Targeted Agents for BRCA/PALB2 and KRAS Mutations PARP inhibitors (olaparib, niraparib) provide a ~2.5‑month OS benefit as maintenance after platinum‑based therapy in germline BRCA/PALB2 patients (POLO trial). KRAS G12C inhibitors (sotorasib, adagrasib) achieve 30‑40 % response rates in early trials; combination with gemcitabine or nab‑paclitaxel improves disease‑control rates. Emerging KRAS G12D inhibitors (MRTX1133, RMC‑6236) are in phase‑I/II studies.
Device‑Based Approaches (Optune Pax Tumor Treating Fields) The FDA‑approved Optune Pax device, combined with gemcitabine‑nab‑paclitaxel, adds ~2 months median OS and improves pain control in locally advanced disease, and investigational trials are extending its use to metastatic settings.
Emerging Metabolic and Stromal Modifiers PRMT5 inhibition (GSK3326595) and metabolic agents (glutaminase inhibitors, autophagy blockers) show synergistic tumor shrinkage with chemotherapy. Stromal‑depleting agents (PEGPH20, FAK inhibitors) enhance drug delivery and modestly extend progression‑free survival.
New Treatments for Pancreatic Cancer Stage 4 Intensified chemo backbones remain foundational, now paired with maintenance PARP inhibition for DNA‑repair‑deficient tumors, KRAS G12C inhibitors for the small G12C subset, and Optune Pax for synergistic tumor‑control. Personalized mRNA vaccines and metabolic‑targeting combos are entering early‑phase trials, offering cutting‑edge options for stage 4 patients.
Best Clinical Trials for Stage 4 Pancreatic Cancer Promising studies include the COMBAT‑PD trial (chemotherapy + pembrolizumab), KRAS‑G12D inhibitor trials, Phase 3 RASolute 302 (RMC‑6236), BXCL701 + pembrolizumab (immune activator), and the APRICOT neoadjuvant trial for unresectable disease. Enrollment in KRAS‑targeted or immunotherapy‑enhancing trials provides access to the most advanced regimens. Learn more about ongoing clinical trials.
New Drug for Pancreatic Cancer Approved by FDA In 2023 the FDA granted accelerated approval to lurbinectedin for KRAS G12C‑mutated metastatic PDAC. The most recent therapeutic approval (Feb 2026) is the Optune Pax device, delivering Tumor Treating Fields with gemcitabine/nab‑paclitaxel for locally advanced disease.
Supportive Care, Lifestyle, and Non‑Conventional Options
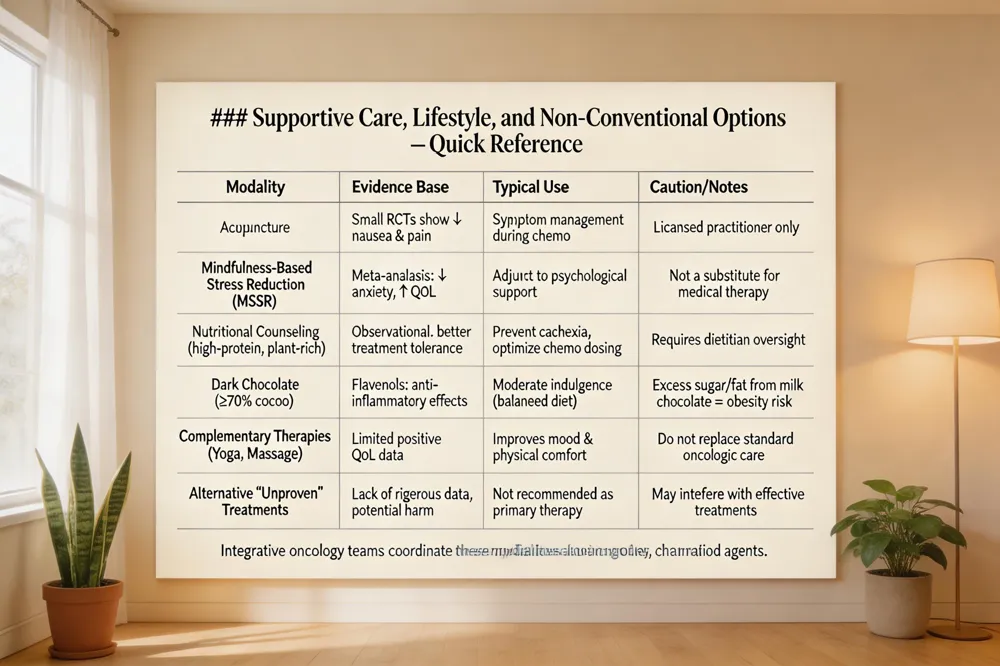 Integrative oncology is now a routine component of pancreatic cancer care in the United States. Multidisciplinary teams—including medical oncologists, nutritionists, psychologists, and acupuncturists—offer evidence‑based complementary therapies such as acupuncture for chemotherapy‑induced nausea, mindfulness‑based stress reduction for anxiety, and targeted nutritional counseling to improve treatment tolerance.
Is chocolate bad for pancreatic cancer? Current data do not link chocolate consumption to tumor progression. Dark chocolate (≥70 % cocoa) contains flavanols with anti‑inflammatory properties and can be part of a balanced, plant‑rich diet. Excessive intake of milk chocolate or sugary candy bars adds calories, saturated fat, and sugar, potentially contributing to obesity—a known risk factor for pancreatic disease. Moderate, occasional dark‑chocolate consumption is unlikely to harm patients.
What is non‑conventional cancer treatment? It refers to therapies outside standard surgery, chemotherapy, radiation, or FDA‑approved targeted agents. Complementary approaches (e.g., acupuncture, yoga, mindfulness) are used alongside conventional care to alleviate symptoms, whereas alternative modalities that replace proven treatments lack robust evidence and may cause harm.
Alternative cancer treatments vs conventional Conventional therapies have demonstrated efficacy through rigorous trials. Complementary modalities can improve quality of life when integrated into an evidence‑based plan, but they should never substitute curative or life‑prolonging treatments.
M.S. cancer clinical trials MD Anderson MD Anderson is recruiting for the EXPAND trial (NCT06593431), a phase III study of multimodal therapy for oligometastatic pancreatic ductal adenocarcinoma, plus several stromal‑targeting, KRAS‑directed, and vaccine trials. Patients can contact 1‑877‑MDA‑6789 for eligibility screening.
Overall, supportive‑care strategies—grounded in integrative oncology, sensible diet, and coordinated multidisciplinary support—enhance patient well‑being while preserving the primacy of evidence‑based cancer therapy.
A Future Fueled by Science, Compassion, and Collaboration
The next era of pancreatic ductal adenocarcinoma treatment hinges on rational combination regimens that pair standard cytotoxic backbones—FOLFIRINOX or gemcitabine + nab‑paclitaxel—with targeted agents such as KRAS G12C inhibitors, PARP inhibitors for BRCA‑mutated disease, or STAT3 degraders. Pre‑clinical triple‑targeted therapy (RMC‑6236, afatinib, SD36) has already produced durable tumor regressions and prevented resistance in mouse models, while early clinical data show that adding KRAS inhibitors to chemotherapy improves disease‑control rates. Hirschfeld Oncology has built a multidisciplinary framework that rapidly translates these trial findings into patient care: molecular profiling guides enrollment in biomarker‑driven studies, and supportive‑care teams manage the complex toxicities of multimodal regimens. Patients are urged to seek clinical‑trial participation, as real‑world evidence demonstrates a 10‑15% survival advantage for those receiving biomarker‑driven combination therapies compared with chemotherapy alone.





.png)


.png)
.png)




