Why Genotype‑Guided Therapy Matters
Pharmacogenomics examines inherited DNA variants that alter drug metabolism, efficacy and toxicity, making it a cornerstone of modern cancer care. In gastrointestinal oncology, germline markers such as DPYD (fluoropyrimidine metabolism) and UGT1A1*28 (irinotecan glucuronidation) predict severe neutropenia, mucositis, diarrhea and hand‑foot syndrome. Genotype‑guided dosing reduces grade ≥ 3 toxicities by up to 50 % while preserving response rates, translating into fewer emergency visits, lower hospitalization costs and improved quality of life. The strategic goal of personalized dose escalation is to start high‑risk patients at reduced doses, then safely increase exposure based on real‑time tolerability and therapeutic drug monitoring, thereby maximizing therapeutic intensity for those with rapid drug clearance and minimizing harm for poor metabolizers.
Pharmacogenomic Testing: Foundations and Clinical Impact
 Pharmacogenomic (PGx) testing examines inherited DNA variants that predict how a patient metabolizes and responds to cancer drugs, allowing oncologists to tailor chemotherapy for maximal efficacy and minimal toxicity. Key germline genes in gastrointestinal oncology include DPYD, which governs fluoropyrimidine clearance; UGT1A1, which affects irinotecan glucuronidation; and CYP2D6, TPMT, which influence other agents. Clinical decision‑support (CDS) alerts embedded in electronic health records automatically flag DPYD and UGT1A1 results and suggest genotype‑guided dose reductions (e.g., 50 % reduction for DPYD intermediate metabolizers, 30‑40 % for UGT1A1*28 homozygotes). This integration shortens trial‑and‑error prescribing and improves adherence. By identifying patients who can safely tolerate higher exposures, pharmacogenomics guides personalized dose escalation, improving response rates while protecting against severe adverse events. Consequently, cancer therapy can be truly personalized—matching tumor biology with patient‑specific metabolism—transforming standard regimens into precision‑driven treatments.
DPYD and UGT1A1: Guiding Safe Dose Escalation
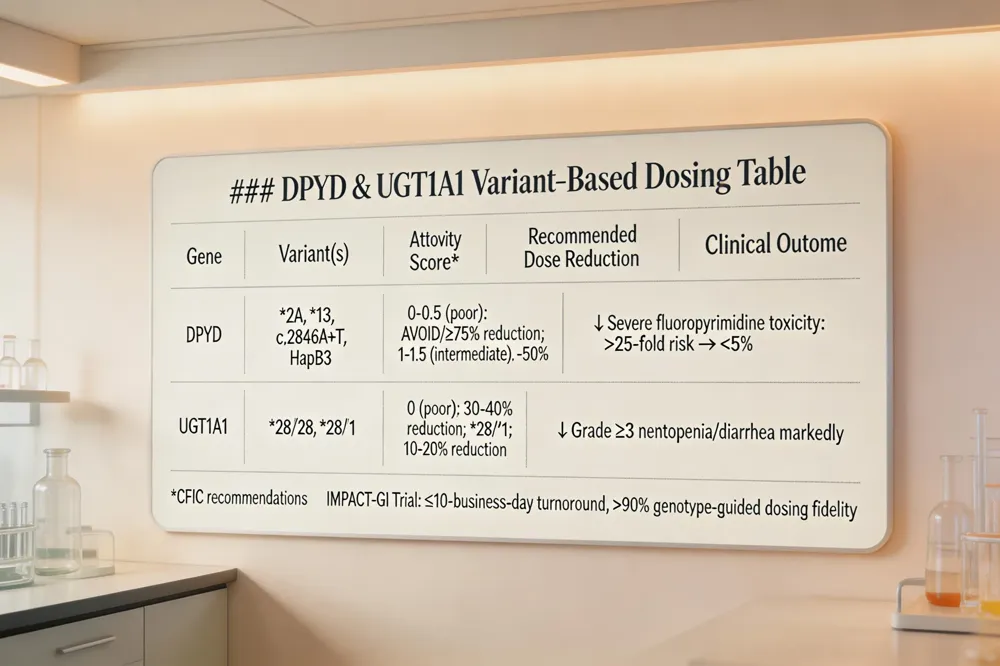 DPYD loss‑of‑function alleles (e.g., 2A, *13, c.2846A>T, HapB3) markedly reduce dihydropyrimidine dehydrogenase activity, raising the risk of life‑threatening fluoropyrimidine toxicity by >25‑fold. CPIC assigns activity scores (normal = 2, intermediate = 1‑1.5, poor = 0‑0.5) and recommends a 50 % dose reduction for intermediate metabolizers and avoidance or >75 % reduction for poor metabolizers. UGT1A128 and 6 polymorphisms impair glucuronidation of irinotecan’s active metabolite SN‑38, increasing severe neutropenia and diarrhea; FDA and CPIC advise 30‑40 % dose reductions for *28/28 homozygotes. The IMPACT‑GI implementation trial, guided by the EPIS (Exploration‑Preparation‑Implementation‑Sustainment) framework, embeds rapid (≤10‑business‑day) DPYD/UGT1A1 genotyping into the Epic EHR, triggering clinical‑decision‑support alerts that enforce genotype‑based dosing before the first chemotherapy cycle.
Side effects of precision medicine include classic chemotherapy toxicities (nausea, fatigue, myelosuppression) when agents affect normal cells, plus modality‑specific adverse events such as skin rash, hypertension, or immune‑related colitis.
Controversy surrounds pharmacogenomics because race is a poor proxy for actionable variants, raising equity concerns, while privacy, consent, and the high cost of genotype‑directed therapies fuel debate over widening versus narrowing access to care.
Precision Medicine in Practice: From Tumor Genomics to Targeted Therapies

Definition and Goals of Precision Oncology
Precision oncology tailors therapy to a patient’s individual tumor genetics, aiming to maximize efficacy while minimizing unnecessary toxicity. By sequencing tumor DNA, clinicians identify actionable alterations and match them to targeted drugs, immunotherapies, or clinical‑trial options.
Actionable Mutations
Key somatic drivers include KRAS, BRAF, HER2 amplification, and microsatellite‑instability high (MSI‑H). These biomarkers guide selection of anti‑EGFR antibodies, BRAF/MEK inhibitors, HER2‑directed therapy, and checkpoint inhibitors respectively.
Tumor‑Agnostic Therapies
Drugs such as larotrectinib and entrectinib target NTRK fusions irrespective of cancer origin, exemplifying a molecular‑first treatment paradigm.
Companies Delivering Personalized Oncology Solutions
Tempus, Guardant Health, Foundation Medicine, and CureMatch provide comprehensive genomic profiling, liquid‑biopsy testing, and algorithm‑driven regimen selection.
Cancer Precision Medicine
Precision medicine in cancer uses a patient’s unique tumor genetics and molecular profile to select the most effective therapies while minimizing unnecessary toxicity. It integrates genomic data with health, lifestyle, and treatment history to improve prognosis and trial enrollment.
Examples of Precision Medicine in Cancer
Patients with HER2 amplification receive trastuzumab; KRAS‑wild‑type colorectal cancer benefits from anti‑EGFR antibodies; MSI‑H tumors respond to pembrolizumab; pharmacogenomic testing guides chemotherapy dosing.
Personalized Cancer Treatment Companies
Tempus, Guardant Health, Foundation Medicine, and CureMatch leverage profiling and data analytics to match patients with optimal therapies.
Precision Oncology for Prostate Cancer
Genomic classifiers (Decipher, Oncotype DX) and liquid‑biopsy assays identify high‑risk disease, guide PARP‑inhibitor use, and support active‑surveillance decisions, enabling truly individualized prostate‑cancer care.
Clinical Trials and Implementation Frameworks
 Design of cancer cure trials focuses on interventions that aim to eradicate disease rather than merely control it. Such trials may employ immunotherapy, targeted agents, novel surgical techniques, or emerging technologies like tumor‑treating fields, enrolling patients early and providing intensive monitoring. A cancer clinical trial is a structured research study that evaluates new preventive, diagnostic, or therapeutic approaches; it proceeds through Phase I (safety and dose), Phase II (early efficacy), and Phase III (comparison with standard care), offering participants access to cutting‑edge treatments while generating data for future practice. Recent cancer‑treatment studies illustrate this trend: tumor‑infiltrating‑lymphocyte therapy shows promise in metastatic gastrointestinal cancers, engineered adipocytes and AI‑driven response tools such as SCORPIO are expanding precision options, and CAR‑T and ecDNA‑targeted CHK1 inhibition are entering early‑phase trials. The IMPACT‑GI implementation study exemplifies a pragmatic, non‑randomized trial that integrates pre‑treatment DPYD and UGT1A1 testing into routine GI oncology. Guided by the EPIS (Exploration, Preparation, Implementation, Sustainment) framework, the trial demonstrated feasibility (results before first dose), fidelity (dose adjustments matching genotype‑based recommendations), and penetrance (high testing rates). Retrospective data from 2017‑2018 showed that ~50% of GI cancer patients experienced severe toxicity, underscoring the real‑world safety impact of genotype‑guided dosing. Together, these elements illustrate how rigorous trial design, phased endpoints, implementation science, and real‑world evidence converge to advance personalized, curative oncology.
Integrating Genomics into Routine Care: Challenges and Opportunities
 Implementing pre‑emptive pharmacogenomic testing for DPYD and UGT1A1 in gastrointestinal oncology faces three primary barriers. First, long turnaround times can delay therapy; recent trials have reduced assay time to ≤10 business days, but even faster (<5 days) is needed for same‑day dosing. Second, seamless EHR integration is essential; embedding discrete genotype data and automated clinical decision‑support alerts in Epic improves fidelity of dose adjustments. Third, reimbursement uncertainty hampers adoption—while many insurers now cover FDA‑approved panels when results affect chemotherapy dosing, variable policies still cause out‑of‑pocket costs for patients. Targeted clinician education—continuing‑education modules for pharmacists and in‑service presentations for physicians—enhances understanding of CPIC dosing guidelines and improves adherence. Parallel patient education addresses genetic literacy, insurance‑discrimination concerns, and the value of informed consent, fostering agency and shared decision‑making.
Can I choose my own oncologist? Patients rarely select oncologists directly; they prioritize experience, reputation, and quality of care over cost, though treatment expenses remain a major concern.
Personalized medicine cancer review Gener PCM uses genomic profiling to match tumors with targeted therapies, improving efficacy and lowering toxicity. Successful rollout requires interdisciplinary leadership, coordinated funding, robust bio‑informatics, and patient‑centered decision‑making.
Precision Oncology journal This open‑access journal publishes high‑impact studies on molecular advances and AI‑driven biomarker discovery, guiding clinicians in translating genomic data into actionable treatment strategies.
Future Directions: Adaptive Dosing, AI, and Emerging Biomarkers
 AI‑driven clinical decision support tools now ingest EHR‑integrated genotype data (DPYD, UGT1A1, TYMS, MTHFR, microRNA signatures) and patient‑specific PK/PD parameters to generate personalized dosing recommendations at the point of care. Machine‑learning algorithms can predict who will tolerate dose escalation or require de‑escalation, streamlining clinician workflow.
The growing role of precision and personalized medicine for cancer treatment – Precision oncology matches each patient’s tumor and germline genomic profile to the most effective therapies, expanding options for pancreatic and colorectal cancers while minimizing unnecessary toxicity.
Can I choose my own oncologist? – Patients prioritize experience, reputation, and quality of care over cost, though insurance considerations remain a concern.
Which role does pharmacogenomics play in cancer treatment quizlet? – Pharmacogenomics informs drug metabolism and toxicity risk, guiding dose reductions for DPYD‑ or UGT1A1‑deficient patients and identifying emerging biomarkers (TYMS, MTHFR, microRNA) that may enable safe dose escalation in the future.
A New Era of Safe, Effective Dose Escalation in GI Oncology
Genotype‑guided dosing transforms gastrointestinal oncology by matching fluoropyrimidine and irinotecan exposure to each patient’s DPYD and UGT1A1 activity. Studies such as IMPACT‑GI and retrospective analyses show a 30‑50 % reduction in grade ≥3 toxicities and fewer hospitalizations, while maintaining response rates. At Hirschfeld Oncology a team of oncologists, pharmacists, genetic counselors and informaticians integrates pre‑emptive testing into the EHR, delivers clinical decision support, and tailors dose escalation or de‑escalation based on CPIC guidelines. This collaborative model ensures safety, preserves dose intensity, and improves quality of life. We call on cancer centers, payers and policymakers to fund universal pre‑emptive panels, embed genotype data in electronic records, and train clinicians, so that precision dosing becomes standard care for all GI cancer patients.





.png)


.png)
.png)




