A patient once asked me, “If treatment is supposed to kill cancer cells, why do some tumors keep acting like nothing happened?” That question gets to the heart of apoptosis and cancer. Some cancers grow not only because they divide too much, but because they stop dying when they should.
An Introduction to Apoptosis and Its Role in Health
Your body is constantly making decisions about which cells should stay and which should leave. That might sound alarming at first, but it’s one of the most protective things your body does.
A useful way to think about it is to picture the body as a busy city. New buildings go up, old ones are repaired, and unsafe structures are taken down before they endanger the neighborhood. Apoptosis is that controlled demolition system at the cellular level. It’s often called programmed cell death, but that phrase can make it sound harsh. In reality, it’s a careful act of maintenance.
Cells use apoptosis when they’re too damaged to fix safely, when they’ve reached the end of their useful life, or when the body no longer needs them. This process helps shape normal development, maintain healthy tissues, and prevent damaged cells from lingering. A cell doesn’t explode or spill chaos into surrounding tissue. It shuts down in an orderly way, and nearby cleanup cells remove the remains.
Apoptosis is less like an accident and more like a planned retirement with safe cleanup afterward.
This matters in cancer because a healthy body depends on the ability to remove dangerous cells early. If that removal system weakens, damaged cells can stay alive, collect more mistakes, and eventually behave like tumors.
For patients with advanced or resistant cancer, this isn’t just biology class. It helps explain why one treatment may stop working, why another may work unexpectedly well, and why newer therapies increasingly aim not only to attack cancer directly, but to restore the cell’s ability to die when it should.
The Body's Essential Cleanup Crew Understanding Apoptosis
Apoptosis follows a sequence. It isn’t random, and it isn’t messy. When it works well, a cell receives a signal, turns on a set of internal enzymes, dismantles itself into small sealed fragments, and gets cleared away without inflaming nearby tissue.
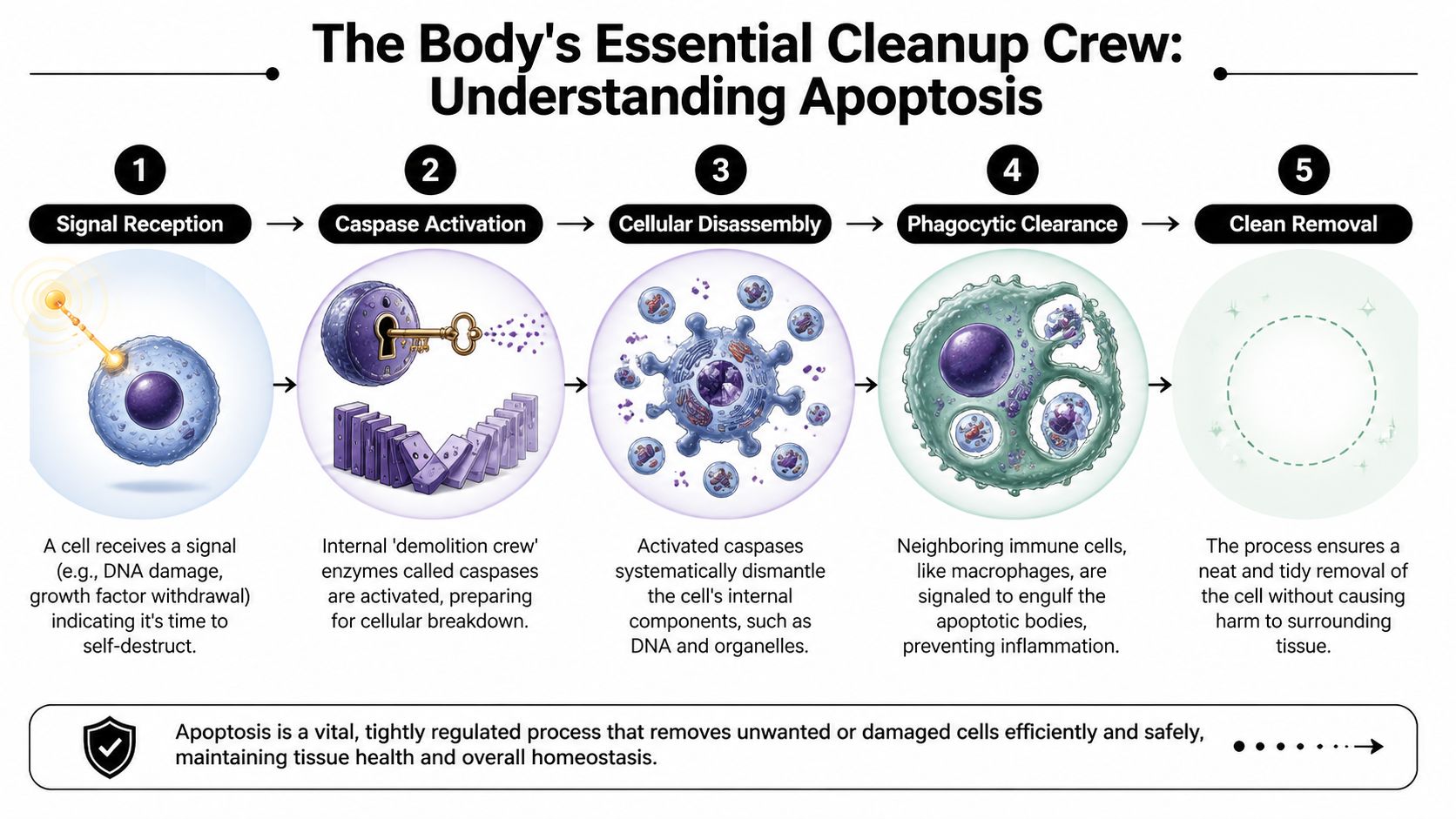
Two ways the process begins
Doctors and scientists usually describe apoptosis as starting through two main pathways. The names sound technical, but the basic idea is simple.
The first is the intrinsic pathway. This is the cell responding to trouble from within. DNA damage, severe internal stress, or loss of key survival support can trigger it. In this pathway, the cell communicates, “Something is wrong inside me, and I may be unsafe to keep.”
The second is the extrinsic pathway. This starts when signals from outside the cell tell it that it’s time to shut down. Other cells in the body, including immune cells, can send those signals. In plain terms, this is more like receiving an external order.
Both pathways end in the same place. They activate a family of enzymes called caspases.
Caspases are the demolition workers
If apoptosis is a demolition project, caspases are the workers who carry it out. Once activated, they begin cutting apart the cell’s structural and regulatory parts in a very organized order.
That organization matters. The cell membrane changes in a way that attracts cleanup cells. The DNA is broken down. Internal parts are packaged into small fragments often called apoptotic bodies. Then scavenger cells, especially macrophages, engulf and remove the remains.
Here’s the key distinction that confuses many readers: apoptosis is not the same thing as uncontrolled cell injury.
- Apoptosis is orderly: The cell dismantles itself with minimal disruption to nearby tissue.
- Necrosis is chaotic: A damaged cell swells, ruptures, and spills contents that can irritate surrounding tissue.
- Why that matters: Orderly removal helps the body maintain balance. Chaotic injury can provoke inflammation and collateral harm.
The mitochondria are more than power plants
Many patients have heard of mitochondria as the cell’s energy centers. In apoptosis, they also act like a decision hub. When the intrinsic pathway is triggered, proteins at the mitochondrial membrane help determine whether the cell will survive or die.
Some proteins push the cell toward death. Others block that decision. Cancer often exploits that balance, which is why drugs that target these proteins have become such an important area of oncology.
Practical rule: When your oncologist talks about “cell death pathways,” they’re often referring to the molecular switches that decide whether damaged cancer cells can still be removed.
Why healthy tissues need this system
Apoptosis protects the body in ordinary life, not just during illness. Immune cells that are no longer needed can be removed. Cells with damaged DNA can be eliminated before they become a bigger problem. Tissues can renew themselves without piling up faulty parts.
That’s why apoptosis and cancer are so closely linked. Cancer doesn’t only involve too much growth. It also involves a failure of restraint. A dangerous cell survives long enough to keep dividing.
A helpful way to summarize the process is this:
| Stage | What happens |
|---|---|
| Signal received | The cell senses internal damage or receives an outside death signal |
| Decision made | Internal proteins determine whether survival is still safe |
| Caspases activate | The cell turns on enzymes that execute the shutdown program |
| Cell breaks apart | Contents are packaged into controlled fragments |
| Cleanup occurs | Macrophages and other cells remove the remains |
When patients understand this sequence, many treatment decisions start to make more sense. A drug may damage a cancer cell. But damage alone isn’t always enough. The cancer cell also has to respond to that damage by entering apoptosis. If it can’t, treatment may look active on paper but disappoint in real life.
The Link Between Faulty Apoptosis and Cancer Growth
One of the most important guardians of this process is p53, a tumor suppressor gene that acts like a quality control inspector. When a cell’s DNA is damaged, p53 helps decide what happens next. It can pause the cell cycle, support repair, or direct the cell toward apoptosis if the damage is too severe.
When that safeguard is lost, the consequences are serious. Defects in the p53 tumor suppressor gene have been linked to more than 50% of human cancers according to this review of p53 and apoptosis in cancer therapy. The same review explains that when p53 is mutated, its protective functions are inactivated, damaged cells survive, and the transcription of pro-apoptotic genes is impaired. That makes cells less able to undergo programmed death and more resistant to chemotherapy and radiotherapy.
Why p53 matters so much
A healthy p53 system asks hard questions before a cell is allowed to keep dividing:
- Is the DNA intact?
- Can this damage be repaired safely?
- If not, should this cell be removed?
When p53 is working, it prevents many risky cells from getting farther down the road. When it’s mutated, that checkpoint weakens.
The result isn’t just faster growth. It’s the survival of cells that should have been stopped early. Those cells can collect additional abnormalities, become harder to control, and respond poorly to treatments that depend on DNA damage to trigger cell death.
Why treatment can fail even when it reaches the tumor
Chemotherapy and radiation often work, in part, by creating enough damage that a cancer cell should recognize it can’t safely continue. That recognition step is essential.
If p53 is defective, the cell may not respond normally. It may absorb damage without fully activating the self-destruct program. From a patient’s perspective, this helps explain a frustrating experience: a scan, biopsy, or treatment plan can look appropriate, yet the tumor behaves as though it has found a workaround.
A resistant cancer cell is often not ignoring treatment completely. It may be receiving the insult but failing to convert that insult into apoptosis.
A simple analogy
Think of p53 as a building inspector with authority to condemn unsafe property. If the inspector is absent, bribed, or unable to file the report, a damaged structure stays open. People keep using it. More problems accumulate. Eventually the risk spreads beyond the original defect.
That’s the bridge between apoptosis and cancer growth. Cancer cells often survive because the body’s internal “remove this now” system has been disabled.
For patients, this understanding can be clarifying instead of discouraging. It means resistance isn’t always mysterious. In many cases, it reflects a specific biological weakness in the tumor’s control system. Once that weakness is identified, doctors can start asking better questions about whether treatment should focus on direct damage, on restoring death signaling, or on bypassing the broken pathway entirely.
How Cancers Learn to Evade Apoptosis
Cancer doesn’t just benefit from a damaged safety system. It often develops active methods to protect itself. A useful way to picture this is to think of the cancer cell as a fugitive who has learned how to disable alarms, block incoming calls, and reinforce every exit.

One major strategy involves Bcl-2, an anti-apoptotic protein. A review on apoptosis resistance and Bcl-2 family signaling notes that increased Bcl-2 expression correlates with worse malignancy prognosis and is directly related to chemoradiotherapy resistance. The same review explains that Bcl-2 prevents the release of cytochrome c from mitochondria, and that the ratio of anti-apoptotic proteins like Bcl-2 to pro-apoptotic proteins like Bax helps determine whether a cell lives or dies.
The cancer cell presses the brake
In healthy tissue, there’s a balance between proteins that promote apoptosis and proteins that restrain it. Cancer often tips that balance toward survival.
That shift can happen in several ways:
- More anti-apoptotic protection: The cell produces stronger internal blockers such as Bcl-2.
- Less pro-death pressure: The signals that would normally push the cell toward apoptosis become weaker.
- A changed survival threshold: The tumor can tolerate levels of stress or treatment damage that would kill a less protected cell.
This is one reason treatment resistance can emerge over time. A therapy may still reach the tumor, but the tumor has altered the machinery that decides whether injury becomes cell death.
Some cancers also stop listening
A second strategy is to reduce sensitivity to outside death signals. On the cell surface, receptors act like communication antennas. If those receptors are reduced or if the downstream signal is blocked, the cancer cell becomes harder to command from the outside.
Pancreatic and other resistant tumors may weaken these surface pathways while also strengthening internal anti-apoptotic defenses. That combination makes them difficult opponents. They’re not relying on one escape route. They’re building several.
If you’re facing this after standard treatment, this overview of what happens when chemotherapy stops working can help frame the next discussion with your care team.
Why this changes the treatment conversation
When people hear “the cancer is resistant,” they often imagine the drug was too weak. Sometimes the deeper issue is different. The cancer has become better at surviving the same damage.
That changes what an oncologist looks for. Instead of asking only, “How can we hit the tumor harder?” we may ask:
| Evasion tactic | What it means clinically |
|---|---|
| Anti-apoptotic proteins rise | The cell resists internal death signals |
| Death receptor signaling weakens | The cell becomes less responsive to external kill signals |
| Multiple defenses stack together | Single-agent treatment is less likely to work |
For many patients, that shift is important. It moves the conversation from blame or hopelessness to mechanism. A tumor that evades apoptosis is revealing something about how it survives. That information can guide more targeted combinations rather than endless escalation of toxicity.
Reawakening Apoptosis Modern Cancer Therapies
Once we understand how cancer blocks apoptosis, the next step becomes more logical. We try to remove the block, bypass it, or trigger a different death signal the tumor can’t ignore.
This is one of the most hopeful areas in modern oncology. Instead of using only broad damage, many newer strategies are designed around the exact points where the cancer has interrupted cell death.
Blocking the proteins that keep cancer alive
Some therapies focus on anti-apoptotic proteins, especially within the Bcl-2 family. These treatments aim to release the brake that cancer has placed on cell death.
A familiar example in oncology is the broader class of BH3 mimetics, which are designed to interfere with survival proteins such as Bcl-2. In plain language, these drugs try to reopen the door to mitochondrial apoptosis. They do not merely poison the cell. They change the internal decision-making environment so the damaged cell is more able to die.
That approach matters most when a tumor’s survival machinery is doing much of the protective work. In those settings, a targeted apoptosis strategy can make a resistant cancer more vulnerable again.
Disabling IAPs and restoring caspase activity
Another set of targets involves IAPs, or inhibitor of apoptosis proteins. These proteins interfere with caspase activity, which means they obstruct the very enzymes that carry out the death program.
According to this discussion of apoptosis resistance and treatment strategies in solid tumors, overexpression of IAPs and Bcl-2 family members in breast, ovarian, and colorectal cancers blocks caspase activation, contributes to immune evasion, and is associated with poor prognosis. The same source states that recent studies from 2025-2026 describe certain natural compounds that can upregulate Bax and p53 while downregulating Bcl-2, inducing significant apoptosis in cancer cell lines and showing synergy with PD-1 inhibitors. Because those years are future-dated within the source framing, it’s best understood as an emerging research direction rather than established standard practice.
Here’s a short visual summary of the main therapy categories clinicians may discuss:
| Therapy Type | Mechanism of Action | Example Target |
|---|---|---|
| BH3 mimetics | Block anti-apoptotic proteins to restore intrinsic apoptosis | Bcl-2 family proteins |
| SMAC mimetics | Counter IAP-mediated suppression of caspases | IAP proteins |
| TRAIL-directed agents | Stimulate external death receptor signaling | TRAIL receptors |
| Combination immunotherapy approaches | Help immune attack succeed when apoptotic resistance is part of the problem | PD-1-related resistance patterns |
Pushing the extrinsic pathway
Some therapies try to activate death signaling from the outside of the cell. This includes efforts to engage TRAIL receptors, which can trigger the extrinsic apoptosis pathway.
In pancreatic cancer research, TRAIL-related approaches are particularly interesting because some tumors may still retain parts of the downstream death machinery even when they resist standard therapy. That doesn’t mean every patient will benefit, and it doesn’t mean these strategies work in isolation. It means oncologists can sometimes test whether an apparently resistant tumor still has an exploitable vulnerability.
Why combinations matter
A tumor may block apoptosis at more than one point. That’s why combination therapy often makes more sense than a single elegant drug used alone.
One treatment may weaken anti-apoptotic proteins. Another may improve immune recognition. A third may create a level of cellular stress that the cancer can no longer buffer. Together, these approaches can do what none of them could do on their own.
For patients trying to understand how immunotherapy fits into this picture, this guide to what immunotherapy for cancer involves is a useful companion.
After the science, it helps to see the idea visually.
How this shows up in real treatment planning
In practice, apoptosis-directed therapy isn’t one single protocol. It’s a way of thinking about cancer biology.
An oncologist may ask:
- What survival proteins are active in this tumor? If anti-apoptotic signaling is dominant, a targeted inhibitor may be relevant.
- Is immunotherapy failing because tumor cells still can’t execute death? In some patients, the immune system may recognize cancer but still struggle to eliminate it.
- Would a lower-toxicity combination make more sense than dose escalation? Especially in advanced disease, the goal is often to preserve function while exploiting a specific weakness.
At centers that treat complex or resistant disease, including practices such as Hirschfeld Oncology, this may translate into personalized combinations of immunotherapy, targeted therapy, and lower-dose chemotherapy strategies chosen for both biologic rationale and tolerability.
Clinical perspective: The key question isn’t only whether a drug can damage cancer. It’s whether the tumor still has a path to convert that damage into meaningful cell death.
That’s why apoptosis and cancer research has become so clinically relevant. It gives patients and oncologists a more precise map. When standard treatment stops working, the next step may not be “more of the same.” It may be a smarter attempt to reopen a blocked death pathway.
Why This Matters for Advanced and Resistant Cancers
For someone living with advanced cancer, molecular language can feel distant from daily reality. But apoptosis isn’t an abstract topic when a tumor has stopped responding. It can explain why a cancer has become hard to control and why a different treatment strategy may still be reasonable.
A strong example is pancreatic ductal adenocarcinoma, or PDAC. According to this review of apoptotic pathways in pancreatic ductal adenocarcinoma, PDAC has a 5-year survival rate below 5% and shows exceptional resistance to apoptosis. The review describes multi-layered defenses, including reduced death receptor formation and increased anti-apoptotic proteins such as Bcl-2 and Bcl-xL. It also explains why standard cytotoxic therapy may trigger some apoptosis signals at the molecular level yet still fail clinically.
Why brute force often isn't enough
Patients with resistant disease sometimes hear treatment framed as a question of intensity. More drugs. Higher doses. Faster escalation.
But resistant cancers often aren’t merely under-treated. They may be biologically shielded. If the pathways that permit apoptosis are blocked at several checkpoints, extra damage alone may not produce the result you want. It may increase side effects without fully solving the core problem.
That’s why combination strategies are so important in advanced disease. The idea is to identify the barriers that matter most in a given tumor, then choose treatments that can dismantle or bypass those barriers.
PDAC shows the problem clearly
Pancreatic cancer is a powerful example because it often resists both internal and external death signals. It can become less responsive to receptor-driven death commands while also reinforcing internal mitochondrial survival programs.
That has two practical implications:
- A disappointing response doesn’t always mean the treatment choice was irrational. The biology may be unusually defensive.
- Next-line therapy should be guided by mechanism whenever possible. The more we understand about the tumor’s survival tactics, the more rational the next move becomes.
Advanced cancer care often works best when it shifts from “How hard can we hit this?” to “What is this tumor specifically doing to stay alive?”
Why patients should care about this science
If you have stage 4 disease, recurrent disease, or cancer that has progressed after standard therapy, apoptosis research gives you something concrete. It gives language for resistance. It gives a framework for asking better questions. It gives a reason to look at targeted combinations, immunotherapy pairings, and other approaches that aim to restore vulnerability rather than intensify toxicity.
That doesn’t mean every patient will have a matching therapy available right now. It does mean there’s a scientific basis for second opinions, molecular review, and personalized planning.
For many families, that’s a meaningful shift. It turns “nothing worked” into a more useful question: what blocked the cancer cell from dying, and can that block be challenged?
Navigating Your Treatment Journey Questions and Resources
By the time patients learn about apoptosis and cancer, they’re usually trying to make real decisions. The most helpful next step is often not memorizing more terminology. It’s knowing what to ask.
Which questions should I bring to my oncologist
Start with questions that connect directly to your own tumor biology and treatment history.
- Ask about resistance patterns: “Do we know why my cancer may not be responding?” This invites a discussion about whether the tumor may be evading apoptosis, avoiding immune attack, or doing both.
- Ask about molecular testing: “Has my tumor been tested in a way that could guide targeted or combination treatment?” This can open the door to discussing gene changes, protein expression, and whether prior biopsy material is still informative.
- Ask about treatment goals: “Are we trying to shrink the tumor, slow progression, improve symptoms, or all three?” Clear goals help you evaluate whether a proposed regimen fits your priorities.
- Ask what the next treatment is trying to change: The answer may involve apoptosis pathways, immune signaling, or a different vulnerability entirely.
How do I know whether apoptosis-targeting therapy is relevant
Not every cancer patient will receive a drug explicitly labeled as an apoptosis therapy. In practice, the concept shows up in treatment design more broadly.
You may be in this territory if:
- Standard treatment stopped working: Resistance often prompts a closer look at the tumor’s survival mechanisms.
- Your doctor is discussing combinations: That may reflect an effort to overcome more than one defense at once.
- You’ve been told the cancer is biologically aggressive or treatment-resistant: These are the settings where apoptosis-focused reasoning often matters most.
A practical conversation can sound like this: “Do you think my cancer is failing to die in response to treatment, and if so, are there therapies that might reopen that pathway?”
Where can I look for trials and practical support
Clinical trials matter because many apoptosis-related strategies are still evolving, especially in solid tumors and heavily pretreated disease. A useful starting point is this guide to cancer clinical trials, which can help patients and caregivers think through eligibility, logistics, and what trial participation involves.
Families also need support around symptom management and recovery expectations for procedures that may be part of a broader cancer plan. If your care team is discussing local therapies such as ablation, a practical overview of tumor ablation recovery time can help you prepare for the day-to-day side of treatment decisions.
What should caregivers listen for during appointments
Caregivers can be invaluable when the information gets dense. It helps to listen for a few key phrases:
| What you hear | What it often means |
|---|---|
| “Resistance” | The tumor may have adapted biologically, not just progressed randomly |
| “Combination therapy” | The doctor may be trying to overcome multiple survival mechanisms |
| “Targeted approach” | Treatment is being matched to a pathway, mutation, or protein pattern |
| “Tolerability” | The plan is considering quality of life as well as anti-cancer effect |
One final point matters. Patients often feel pressure to make fast decisions while exhausted. You’re allowed to ask for plain language. You’re allowed to ask whether the treatment is intended to attack the tumor directly, help the immune system, or restore the cancer cell’s ability to die.
“Can you explain what this treatment is trying to do inside the cancer cell?” is one of the most useful questions in all of oncology.
If you’re weighing options for advanced or treatment-resistant cancer, Hirschfeld Oncology offers educational resources and consultation pathways focused on personalized regimens, including immunotherapy, targeted therapy, and lower-toxicity combination approaches that can be discussed in the context of your tumor biology and treatment goals.





.png)


.png)
.png)




