Cancer clinical trials are simply research studies designed to find new and better ways to treat, find, and prevent cancer. They are the engine of all medical progress in oncology, and they are how every new treatment gets discovered.
What Are Cancer Clinical Trials, Really?
Think of it this way: standard cancer care is like a map of well-paved, reliable roads. These are the treatments that have been proven safe and effective for many people over time. A cancer clinical trial, on the other hand, is like getting access to a brand-new express lane that’s just been built, based on the very latest engineering and traffic data. It offers a potentially faster, more direct route to your destination.
It’s a common misconception that trials are only a "last resort." For many patients, they are a powerful and proactive first choice, offering access to tomorrow's medicine, today.
A Partnership in Medical Progress
When you join a clinical trial, you're not just a patient; you become a partner. It’s a true collaboration between you, your oncology team, and the researchers working tirelessly to move medicine forward. Every single breakthrough we have in cancer care—from the first chemotherapy drugs to modern immunotherapies—started as an idea tested in a clinical trial.
By participating, you're doing two incredible things at once. You gain access to promising new treatments for yourself, and you contribute to a legacy of knowledge that will help countless people who come after you. You become an active part of the solution.
Moving Beyond Standard Care
For anyone dealing with a complex or advanced cancer, clinical trials can open doors to treatments that won't be widely available for years. Standard protocols are based on what has worked best for large groups of patients in the past. Trials are focused on what might work even better in the future.
This gives you a chance to access innovative approaches, including:
- Targeted Therapies: These are smart drugs engineered to attack specific weaknesses in cancer cells while sparing most healthy tissue.
- Immunotherapies: Treatments that essentially take the brakes off your own immune system, unleashing it to recognize and fight cancer.
- Novel Drug Combinations: Testing new combinations of treatments that, when used together, may be far more powerful than when used alone.
“There are many potential benefits to participating in a clinical trial, including being part of finding new drugs or combination of drugs to treat cancer.” - Jeffrey Meyerhardt, MD, MPH, Chief Clinical Research Officer at Dana-Farber.
Taking Control of Your Journey
A cancer diagnosis can make you feel like you’ve lost control. Exploring all your options, including clinical trials, is a powerful way to get some of that control back. It’s about arming yourself with information and looking at every possible path forward with your doctor. It’s an act of hope.
Every trial is built on a strict foundation of patient safety and ethical rules. The entire process is designed to be transparent and keep your well-being as the absolute top priority. By understanding how they work, you can have more productive conversations with your oncologist and make decisions that feel right for you and your goals. If you'd like to dive deeper, you can find in-depth information about how trials are structured.
Considering a clinical trial means you're leaving no stone unturned. You're ensuring you have access to the very forefront of medical science, where new hope is constantly being created.
Understanding The Four Phases of a Clinical Trial
When you hear the term “cancer clinical trial,” it’s easy to imagine one single, massive experiment. But that’s not quite how it works. In reality, every new treatment moves through a careful, multi-stage process. Each stage, or phase, is designed to answer a different set of questions, building on the knowledge gained from the one before.
Think of it like this: you wouldn't build a roof before the walls are up, and you can't put up walls without a solid foundation. Clinical trial phases work the same way. This methodical approach is the bedrock of medical progress, ensuring that by the time a new therapy is approved, we know it's both safe and effective.
For you as a patient, understanding these phases is empowering. It demystifies the process and helps you ask the right questions when discussing trial options with your care team.
This flowchart shows how a clinical trial can fit into your treatment journey, sometimes offering a more direct path to the latest medical science.
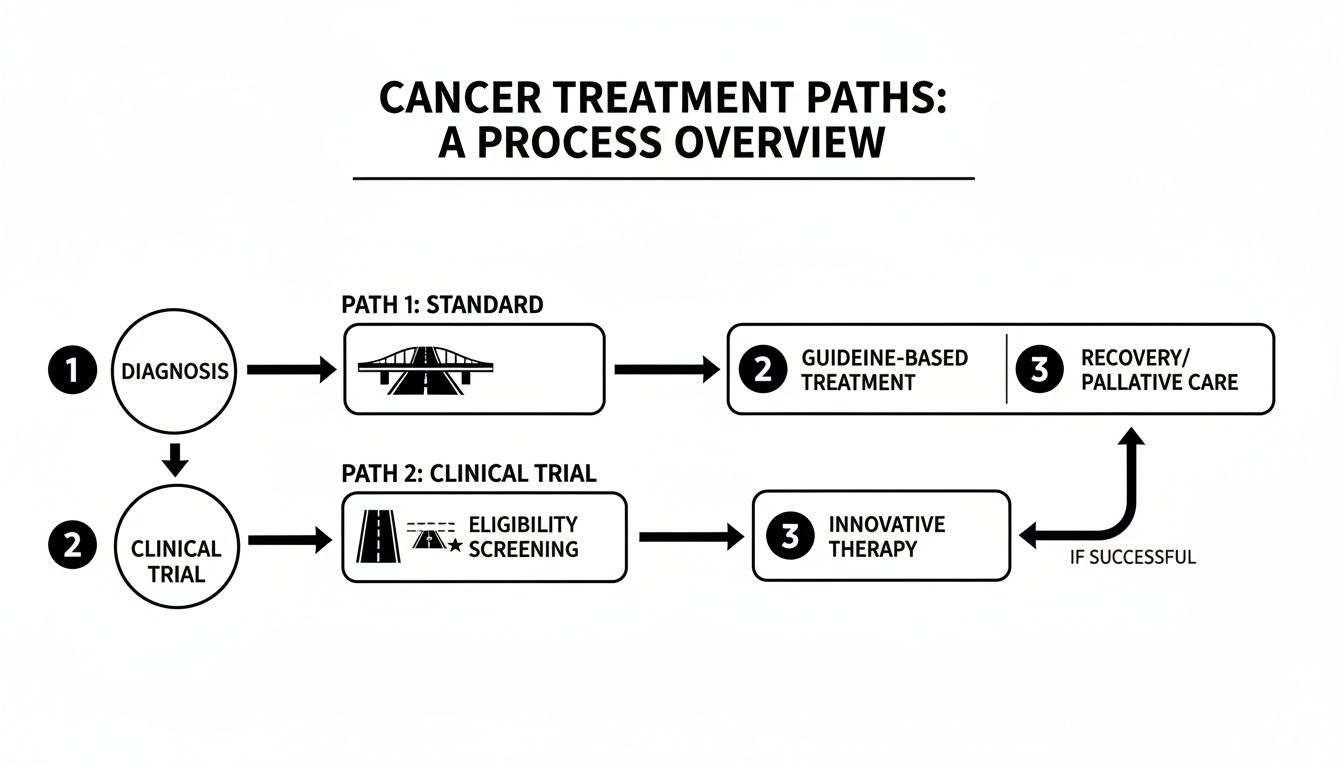
While standard care follows a well-established route, a clinical trial can be an opportunity to access an innovative therapy sooner.
To help you understand what happens at each step, here is a quick overview of the four phases of a clinical trial. Each one has a distinct purpose, a different number of participants, and a specific question it aims to answer.
This table gives you a bird's-eye view, but let's take a closer look at what each phase means in practice.
Phase I: Is It Safe?
Phase I trials are the first step in testing a new treatment in humans. At this point, the primary focus isn't on curing cancer—it’s all about safety. Researchers need to figure out how the human body reacts to the new drug.
These studies involve a very small group of people, often between 15 and 30. The trial starts with a very low dose of the drug, which is then gradually increased in new participants. The goal is to find the highest dose that can be given without causing unacceptable side effects. This process is called dose escalation, and it establishes the groundwork for all future studies.
Phase II: Does It Work?
Once a safe dose has been determined in Phase I, the research can move on to Phase II. The big question now becomes: does this treatment actually work against a specific cancer?
In this phase, the drug is given at the dose identified in Phase I to a larger group of patients, usually all of whom have the same type of cancer. This is where researchers get their first real glimpse of the drug’s effectiveness. They're looking for signs that the treatment is shrinking tumors or stopping cancer from growing. If the results are promising, the drug has a good chance of advancing.
Phase III: Is It Better?
This is the make-or-break moment for a new treatment. A Phase III trial is a large-scale study, often involving hundreds or even thousands of patients at multiple hospitals, designed to answer one crucial question: Is this new treatment better than the current standard of care?
To get a clear answer, patients are usually assigned to different groups at random. One group receives the new treatment, while the other group (the control group) receives the best treatment currently available. This direct comparison is the most rigorous way to prove a new therapy's value. It’s a high bar to clear—only about 25-30% of treatments that enter clinical trials successfully complete Phase III and gain approval from the FDA.
Phase IV: What Else Can We Learn?
The learning process doesn’t end when a drug is approved and becomes available to the public. Phase IV trials, also known as post-marketing surveillance, continue to monitor a drug's safety and effectiveness over the long term.
These studies can involve thousands of people and are crucial for spotting rare or long-term side effects that might not have appeared in smaller, shorter trials. Phase IV research can also explore whether the drug might be useful for other types of cancer or in different combinations, ensuring that our understanding of a treatment continues to evolve for years to come.
The Fast-Paced World of Cancer Research
If you’re exploring cancer treatment options today, it’s important to know that you’re looking at a field that is moving at an incredible speed. The world of oncology isn't static. New discoveries are constantly reshaping what’s possible, bringing more effective treatments—and more hope—to light.
This isn't happening by chance. It’s the direct result of major investments and huge leaps in technology that let scientists see cancer with stunning new clarity. We can now understand tumors at the molecular level, which has opened the door to a new generation of cancer clinical trials focused on treatments designed for the individual, not just the disease.
These smarter, more personalized studies are becoming more common everywhere, but especially in major research centers like New York City, where patients often have first access to the latest breakthroughs.
Precision Medicine: The Driving Force Behind New Treatments
One of the most significant changes in cancer care is the shift to precision medicine. It’s a simple but powerful idea. Instead of treating all lung cancers or all breast cancers the same way, we can now analyze a tumor's unique genetic fingerprint to find its specific weaknesses—its Achilles' heel.
Think of it like having a custom-made key for a specific lock, rather than using a one-size-fits-all master key. This has led to entirely new classes of drugs that are changing the game.
- Targeted Therapies: These are drugs engineered to shut down the specific pathways that cancer cells use to grow and multiply.
- Antibody-Drug Conjugates (ADCs): You can think of these as “smart bombs.” They combine a highly specific antibody that seeks out cancer cells with a powerful chemotherapy drug, delivering the treatment directly to the target while sparing healthy cells.
- Immunotherapies: These treatments don't attack the cancer directly. Instead, they unleash the power of your own immune system, teaching it to recognize and destroy cancer cells.
Because of this focus on personalized medicine, today's cancer clinical trials are much more strategic. The old model of one-size-fits-all oncology is being replaced by a more intelligent, targeted approach. To get a better sense of what these new therapies involve, you can read more about the recent advances in oncology clinical trials.
Following the Investment in Hope
This explosion in new trial options is directly fueled by a massive global effort to find better ways to treat cancer. The sheer amount of money and resources being poured into oncology research is a powerful sign of this commitment.
The global market for oncology clinical trials was valued at $14.04 billion in 2024 and is on track to hit $19.22 billion by 2029.
That growth isn't just a number on a spreadsheet. It represents a worldwide demand for better therapies, and it directly funds the development of the very precision treatments that offer new hope for patients with complex or hard-to-treat cancers. For a deeper dive into these trends, you can explore the full market research report.
For patients and their families, this investment translates directly into real, tangible hope. It means more studies are being launched, more new drugs are being tested, and the scientific community is better equipped than ever to find answers. When you consider a clinical trial, you're not just choosing a treatment—you're stepping into a field that is actively and successfully pushing the boundaries of what's possible in cancer care.
Weighing the Benefits and Risks of Trial Participation

Thinking about a cancer clinical trial brings up a lot of questions. It’s a big decision, and it's completely normal to feel a mix of hope and uncertainty. The key is to get a clear, realistic picture of what joining a trial actually involves so you can weigh the potential upsides against the real-world downsides for yourself.
This isn't just a medical decision; it's a personal one that impacts your health, your time, and your life. Let's walk through what you should be thinking about.
The Potential Upsides of Participation
For many people, the most powerful draw is getting access to the very forefront of medical science. Joining a trial can give you the chance to receive a promising new treatment, sometimes years before it becomes widely available to other patients.
This usually means your care is led by top oncologists who are deeply invested in research. Participants in trials are monitored with incredible care and attention. Those extra check-ups and the close watch from the research team can be a huge source of reassurance for both you and your family.
Of course, the benefits extend beyond your own treatment. When you participate in a trial, you're making a powerful contribution to medicine itself. You become a partner in the research, helping scientists find the answers needed to create better treatments for the future.
In short, the key benefits often include:
- Access to New Treatments: You might be one of the first people to benefit from a new therapy, like a targeted drug or immunotherapy, that isn't available yet.
- Expert Medical Care: Trials are often run at leading cancer centers by physicians who specialize in your exact type of cancer.
- Close Monitoring: A dedicated research team, including nurses and coordinators who know the trial inside and out, will be paying extra attention to your progress.
- Contributing to Science: Your involvement helps push cancer research forward, potentially improving care for thousands of people down the road.
Understanding the Potential Risks
Now for the other side of the coin. It’s just as important to have a frank conversation about the potential risks and uncertainties. By its very nature, a clinical trial is an experiment. The new treatment being studied hasn't been fully proven yet, and that comes with some unknowns.
The most immediate concern for many patients is the risk of side effects. A new therapy can cause side effects that are different, or possibly more severe, than those of standard treatments. While your safety is always the number one priority, this is a real possibility to consider.
There's also no guarantee that the new treatment will work better than the current standard of care. It's possible it could be less effective, or it might not provide any benefit at all. This is exactly what the trial is designed to figure out.
Your participation in a trial is always voluntary. You have the right to leave at any time for any reason, and your decision will never impact the quality of your future care.
Finally, joining a cancer clinical trial often requires a significant time commitment. You may need more frequent trips to the hospital for treatments, lab work, and follow-up visits than you would with standard care. For anyone, but especially those navigating a busy area like NYC, this requires thinking through the logistics of travel and scheduling.
The Power of Informed Consent
This is where informed consent comes in—and it’s the most important part of this whole process. This isn't just about signing a piece of paper. It's an ongoing conversation between you and the research team. They will walk you through every single detail of the study: its purpose, the procedures involved, the potential benefits, and all the known risks.
This is your time to ask anything and everything on your mind. No question is too small. The goal is to arm you with all the information you need to decide if the trial truly aligns with your personal goals and values. It puts the power in your hands to weigh the scales for yourself, so you can move forward with confidence, no matter which path you choose.
How to Find and Evaluate Cancer Clinical Trials

So, you’ve decided to explore cancer clinical trials. Where do you even begin? The internet is a vast and sometimes confusing place, but finding the right study doesn't have to feel like searching for a needle in a haystack.
Your first and most important step should always be a conversation with your oncologist. They know you, your specific diagnosis, and your medical history inside and out. Simply asking, "Is there a clinical trial that might be a good fit for me?" opens the door. They're your best guide for spotting promising studies and helping you decide if a trial’s goals line up with your own.
Once you have that conversation, you can start doing some of your own digging. This makes you an active partner in your own care, and you might even find options to bring back and discuss at your next appointment.
Starting Your Search with Powerful Resources
Thankfully, you don't have to sift through the entire internet. Several reliable, well-organized databases exist to help you find exactly what you're looking for. These platforms let you filter your search by cancer type, trial location, and other critical details.
Here are the best places to start looking:
- ClinicalTrials.gov: This is the big one. Maintained by the U.S. National Library of Medicine, it’s the most comprehensive database out there, listing public and private studies from all over the world.
- National Cancer Institute (NCI): The NCI offers its own searchable database, which often includes patient-friendly summaries that are easier to understand.
- Advocacy Group Websites: Many organizations focused on a specific cancer (like the Pancreatic Cancer Action Network or the Breast Cancer Research Foundation) maintain their own lists of relevant trials for their communities.
When you're using these search tools, the more detail you can provide, the better. Instead of a general search for "cancer," try something more specific like "metastatic pancreatic cancer" and add your city, like "New York City," to see what's available nearby. For those looking into pancreatic cancer options specifically, you can learn more about clinical trial opportunities for pancreatic cancer patients right here on our blog.
Taking the initiative to look for a trial is more relevant today than ever before. Research in oncology is booming—it currently makes up 41% of all clinical trials worldwide. In 2024 alone, there were 2,162 new oncology trial starts, a massive 58% increase over the past decade. A huge part of this growth is in new approaches like cell and gene therapies, which now represent 35% of the oncology pipeline. You can discover more insights about these global oncology trends.
Key Questions to Ask About a Potential Trial
Once you've found a trial that seems promising, it's time to dig into the details. This is where you bring your list back to your oncologist and start asking the tough questions to see if it’s really a good match.
Your goal is to gather enough information to make an informed decision. The answers to these questions will help you and your doctor weigh the potential benefits against the risks and practical commitments.
Critical Questions for Your Care Team:
- What is the main purpose of this study? Is it mainly about safety (Phase I), finding the right dose and effectiveness (Phase II), or comparing a new treatment to the current standard (Phase III)?
- What are the potential benefits and risks? Be direct. Ask for a clear explanation of known side effects as well as the possibility that the treatment may not work.
- What will I be required to do? Get a real sense of the commitment. What is the schedule for treatments, blood draws, and scans? How often will I need to travel to the clinic?
- How will my health be monitored? Close monitoring is one of the key benefits of a trial. Ask what that will look like for you.
- What happens if the treatment works? If you have a good response, is there a way to continue receiving the treatment after the trial ends?
- Will I know which treatment I am receiving? You need to know if the trial involves a placebo or if you could be randomly assigned to different groups (arms).
- Who pays for the costs associated with the trial? Have a frank discussion about finances. What does the trial sponsor cover, and what will be billed to your insurance?
Getting clear answers to these questions is how you move from the abstract idea of a "clinical trial" to a real, tangible option that you, your family, and your doctor can evaluate with confidence.
The Future of Cancer Trials and What It Means for You
If you’re exploring treatment options, it’s important to know that the landscape of cancer clinical trials is changing for the better. We're in an era of incredible discovery, where research is moving faster than ever before. For patients and their families, this means the trials available today—and those on the horizon—are smarter, more targeted, and designed with your quality of life in mind.
The sheer momentum is staggering. By 2025, an estimated 2,174 new oncology clinical trials are expected to launch worldwide. Cancer research now makes up 45% of the entire cell and gene therapy pipeline, a field projected to become an $80 billion market by 2029. With North America at the forefront of this growth, patients here have remarkable access to these studies. You can see a detailed breakdown of these numbers in this full clinical trial trends report.
The Rise of Smarter, More Targeted Therapies
Perhaps the most significant change is the shift from one-size-fits-all treatments to therapies that zero in on a cancer's specific vulnerabilities. Researchers can now identify the unique genetic drivers of a tumor, allowing them to develop treatments that are incredibly precise. This has given rise to a whole new class of therapies being tested in trials.
- CAR T-cell Therapy: This is a fascinating "living drug" approach. We collect a patient’s own immune cells, essentially re-train them in a lab to become expert cancer-fighters, and then infuse them back into the body to hunt down the disease.
- Bispecific Antibodies: You can think of these like a therapeutic matchmaker. These proteins are engineered with two "arms"—one grabs onto a cancer cell while the other grabs an immune cell, forcing them together to initiate a targeted attack.
- Cell and Gene Therapies: This is a wider category of treatments that work by repairing or replacing faulty genes, or by using living cells as medicine. It represents a completely different way of thinking about how to treat cancer.
These aren't just incremental improvements. They represent a new philosophy: instead of just attacking the disease with outside chemicals, we're empowering the body’s own defense systems to win the fight.
Trials That Are More Patient-Friendly
It’s not just the science that’s evolving; the very structure of cancer clinical trials is becoming more accommodating. The focus now is on finding ways to minimize the disruption to your life without ever compromising on safety or the quality of the research.
The entire field is evolving to deliver smarter, kinder, and more personalized care. Innovations are making it easier for patients to access groundbreaking science without completely disrupting their lives.
This patient-first mindset is showing up in a few key ways. Decentralized (or "virtual") trials are on the rise, using tools like video appointments and local lab services to dramatically cut down on travel. At the same time, the increasing use of genomic profiling—where your tumor's DNA is mapped out—helps us match you to a trial based on specific genetic markers. This gives you a much better chance that the treatment being studied will work for you.
Answering Your Questions About Clinical Trials
Even after you understand the basics, a lot of questions can pop up when you’re thinking about a cancer clinical trial. These are big decisions, and getting straight, honest answers is the best way to feel more in control and less anxious. Let’s walk through some of the most common concerns I hear from patients and their families.
Will I Get a Placebo Instead of Real Treatment?
This is probably the number one question people ask, and it’s a completely valid fear. The simple answer is: almost never. In the vast majority of cancer trials, it would be unethical to give you an inactive pill or infusion (a placebo) if a good, standard treatment already exists.
So, how does it work? Usually, a trial compares the new, investigational treatment against the current best standard of care. Sometimes, a placebo is used in addition to standard care. For example, one group of patients might get the standard treatment plus the new drug, while the other group gets the standard treatment plus a placebo. This setup helps researchers know for sure if the new drug is what’s making the difference. You will always be told upfront during the informed consent discussion if a placebo is involved in any way.
Who Pays for the Costs of a Clinical Trial?
I know that figuring out the financial side of treatment can be incredibly stressful. Thankfully, the cost structure for trials is designed to be as clear as possible. The trial sponsor—which is often a pharmaceutical company—pays for all the costs that are purely for research purposes.
This includes things like:
- The investigational drug itself
- Extra doctor visits required only for the study
- Specialized tests or scans that aren't part of routine care
Your own health insurance is generally billed for what’s called "standard of care." These are the costs you would have anyway, even if you weren't in the trial. Before you ever sign on, a research coordinator and financial counselor will sit down with you and map everything out so you know exactly what to expect.
Can I Leave a Clinical Trial After I Have Joined?
Yes. Absolutely. Your participation in any clinical trial is 100% voluntary. You have the right to leave a study at any point, for any reason, and you never have to explain your decision to anyone.
Your choice to withdraw will not negatively affect your relationship with your medical team or the quality of care you receive. If you decide a trial is no longer right for you, your doctors will work with you to smoothly transition to the best available standard treatment plan.
The entire process hinges on clear, constant communication between you and your care team. Building that trust is everything; in fact, understanding proven patient engagement strategies in healthcare shows just how much top-tier research teams prioritize supporting their patients. This focus on respectful dialogue ensures your voice is always heard and your needs are met, whether you stay in the trial or choose another path. Your well-being is always the top priority.
At Hirschfeld Oncology, we believe that empowerment comes from information. Making the best decision for your health means exploring all your options, and our team is here to offer guidance and personalized care every step of the way. To learn more or to schedule a consultation, please visit us at https://honcology.com/blog.





.png)


.png)
.png)




