Before any new cancer treatment can become a standard of care, it must go through a highly structured and meticulous testing process. These are the clinical trial phases, a series of research studies designed to answer one fundamental question at each step: Is this new therapy both safe and effective for patients?
This step-by-step process is the backbone of all medical progress, moving a promising idea from a lab bench to a life-changing treatment.
A Patient's Guide to Clinical Trial Phases
When you're dealing with a cancer diagnosis, the world of clinical trials can seem complicated and even a little intimidating. It’s completely normal to feel that way. But think of these trials not as a last resort, but as a carefully planned journey toward better treatment options.
It's a lot like building a major bridge. You wouldn't just sketch a design and then immediately open it to rush-hour traffic. Engineers follow a strict, phased approach—testing the foundation, the materials, and the structure's integrity under stress—to guarantee it’s safe. Clinical trials work the exact same way, with each phase designed to answer a different, crucial question.
This guide is here to break down that journey for you, especially if you or a loved one is managing an advanced cancer like pancreatic, bile duct, or colorectal cancer. Our goal is to pull back the curtain on this process so you can feel confident and informed.
From a Lab Idea to an Approved Treatment
The path from a scientific discovery to a treatment you receive in the clinic is a long one, but it's logical and built around patient safety. The flowchart below gives you a bird's-eye view of how it all works.
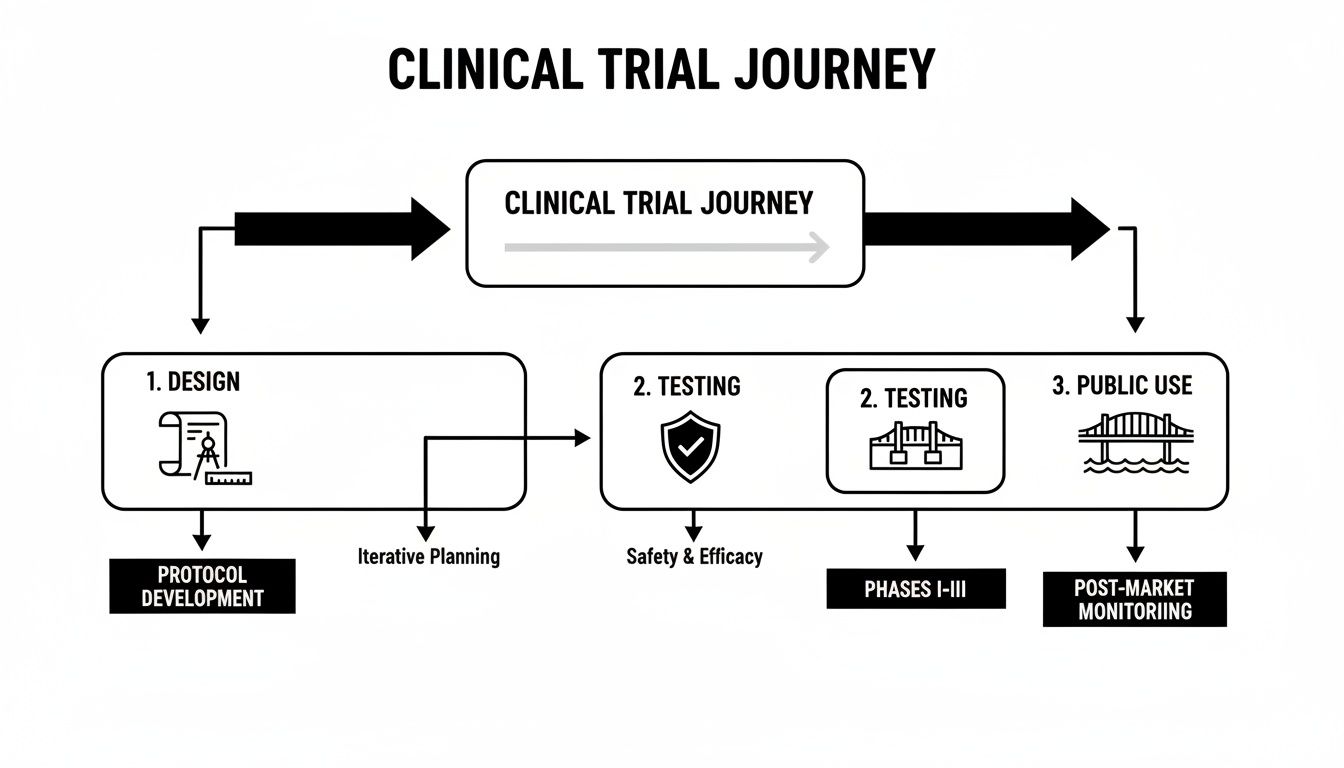
As you can see, each stage builds on the knowledge gained in the one before it. This ensures that by the time a drug is approved, we have a very clear understanding of its benefits and potential risks.
This whole progression is managed by a large network of researchers, doctors, and regulatory experts. If you're curious about the teams working behind the scenes, learning about what Medical Affairs is and why it matters in pharma can give you a fuller picture of how a treatment moves from the lab to your doctor's office.
Every single approved cancer therapy available today exists because patients before you made the courageous choice to participate in a clinical trial. Their involvement is what drives science forward and creates better standards of care for everyone.
Each stage of the trial has a distinct mission:
- Finding a Safe Dose: The very first step is all about safety. Researchers work with a small number of people to find a safe dosage and watch closely for any side effects.
- Checking for Effectiveness: Once a safe dose is established, the next question is: does it actually work against the cancer? This phase focuses on the treatment's impact.
- Comparing to the Standard: Here, the new treatment is compared directly against the current standard therapy in a much larger group of patients to confirm its benefits and see if it's a better option.
- Monitoring in the Real World: Even after a drug is approved, studies continue to track its long-term safety and effectiveness as it's used by the general public.
Understanding the clinical trial phases is the first step toward exploring all your options. To learn more about their impact, you can read our post on how clinical trials are shaping future cancer treatments. This knowledge empowers you to have more meaningful conversations with your oncology team.
The Early Stages: Phase 0, Phase I, and Phase II
Before any new cancer drug can even be considered for widespread use, it has to pass a gauntlet of early-stage tests. These first steps—Phase 0, Phase I, and Phase II—are where a promising idea from the lab first encounters human biology. Each phase is meticulously designed to answer critical questions before the treatment is given to a larger group of people.
It’s a bit like a master chef cautiously working with a rare, powerful new spice. First, they’d test a microscopic speck just to see how it behaves chemically (Phase 0). Then, they’d try a tiny, precisely measured amount to make sure it's safe to eat (Phase I). Only then would they check if the spice actually makes a small batch of the recipe taste better (Phase II).
Phase 0: A First Look in Humans
Not every drug goes through a Phase 0 trial; it's an optional, preliminary step. These studies are tiny, often involving fewer than 15 people. Participants are given a single, sub-therapeutic dose—an amount so small it isn’t expected to have any real treatment effect.
So, what's the point? The goal isn't to fight the cancer but to answer one simple, vital question: does the drug get where it needs to go in the human body? Often called "micro-dosing" studies, these trials allow researchers to see if the drug reaches the tumor and how the body starts to process it.
This quick look can save an enormous amount of time, money, and patient risk. If a drug can't even hit its target in a Phase 0 study, researchers can move on to a more promising candidate without ever starting a larger, more involved Phase I trial.
Phase I: Is It Safe?
This is where the rubber truly meets the road. Phase I is the first time a new treatment is tested in people with the primary goal of establishing its safety. It’s one of the most crucial phases, and patient well-being is monitored with incredible care.
The participant group is still small, usually 20 to 100 individuals. Often, these are patients with advanced cancer who have already been through standard treatment options.
The main questions a Phase I trial seeks to answer are:
- What is the maximum tolerated dose (MTD)? This is the highest dose of the drug that can be given without causing severe or unacceptable side effects.
- What are the side effects? Researchers meticulously log every single reaction, from mild to serious, to build a comprehensive safety profile.
- How does the body handle the drug? This involves tracking how it's absorbed, where it goes, and how it’s eventually cleared from the system.
A Phase I trial isn't really designed to see if the treatment is effective, though everyone is certainly watching for positive signs. The real mission is to build a solid foundation of safety, ensuring the drug is tolerable enough to move forward.
The process is slow and deliberate. The first small group of participants starts on a very low dose. If they tolerate it well, the next small group gets a slightly higher dose. This "dose escalation" continues until researchers pinpoint the MTD.
Phase II: Does It Show Promise?
Once a safe dose has been worked out in Phase I, the treatment advances to Phase II. Here, the focus pivots from safety to the first real test of effectiveness. The big question now is: does this drug actually have a meaningful impact on a specific type of cancer?
Phase II trials are larger, typically enrolling 100 to 300 people who all share the same cancer diagnosis. This is the stage that provides the first real signal of whether a drug has what it takes to one day become a standard therapy.
The core goals of a Phase II trial are to:
- Assess effectiveness. Does the treatment shrink tumors? Does it stop them from growing?
- Continue to monitor safety. With a larger group, researchers can identify less common side effects that may not have appeared in Phase I.
- Confirm the best dose. Is the dose identified in Phase I both safe and effective for this larger population?
This phase is a major hurdle. While oncology clinical trials now drive most global drug development, Phase II is a notorious bottleneck where many promising treatments fail. This is often due to something called 'Phase II signal inflation'—when exciting results in a small, select group of patients simply don't materialize in the larger, more rigorous Phase III trials. You can learn more about the challenges transforming clinical trial development on Clinical Leader.
If a treatment shows enough activity and maintains a good safety record in Phase II, it earns the right to proceed to the final and most demanding stage of testing. This is the make-or-break moment that decides if it gets one step closer to helping patients on a global scale.
The Pivotal Late Stages Phase III and Phase IV
Once a new therapy has proven its basic safety and shown real promise in the early phases, it enters the final, most demanding stages of testing. Phase III and Phase IV trials are what separate a hopeful new drug from a treatment that becomes the standard of care for patients everywhere. These are the large-scale, definitive studies that confirm a treatment's true value on a global stage.

If the earlier trial phases were the qualifying rounds, Phase III is the championship game. This is where the new treatment goes head-to-head with the best treatment already available. It’s the ultimate test to see if a new champion can be crowned.
Phase III Confirming the Winner
The goal of a Phase III trial is simple: get a definite answer. These studies are massive, often enrolling several hundred to over 3,000 participants in clinics and hospitals across different cities, and sometimes different countries. You need that many people from diverse backgrounds to gather the powerful evidence that regulators like the FDA require for approval.
To keep the results fair and unbiased, Phase III trials are almost always randomized. This means patients are assigned by chance to one of two groups:
- The Investigational Group: This group receives the new treatment.
- The Control Group: This group receives the current standard of care.
Many of these trials are also "double-blind," a crucial step where neither the patients nor their doctors know who is getting which treatment. This design might sound strange, but it’s the best way to prevent anyone's hopes or expectations from influencing the results. It ensures the final data is as clean and reliable as it can possibly be. A win in Phase III is what gives a new drug its shot at approval.
Phase IV Real-World Safety and Long-Term Value
But even after a drug is approved by the FDA, the work isn't over. This is where Phase IV begins, often called post-marketing surveillance. Think of it as ongoing quality control, happening out in the real world after a drug is available by prescription.
Phase IV studies can involve thousands of people and can last for many years. They have a few really important jobs:
- Watching for Long-Term Side Effects: They help uncover rare side effects that might only pop up after many more people have used the drug over a longer time.
- Finding New Uses: Sometimes, we learn that a drug is surprisingly effective for other conditions or offers unexpected long-term benefits.
- Optimizing Treatment: Doctors can use these studies to see how the drug stacks up against other options and figure out exactly where it fits into a patient’s overall treatment plan.
By taking part in these later-phase trials, you’re adding to a global well of knowledge. The information gathered helps doctors everywhere make smarter decisions and improves life for patients for generations.
This worldwide approach to research is more important than ever. While North America still leads in the number of trials, the Asia-Pacific region is catching up fast, with its market expected to double by the early 2030s. This global teamwork means that breakthrough therapies tested anywhere in the world can find their way to clinics right here in Brooklyn much faster. To get a better sense of these trends, you can explore the data on global clinical trial success rates from CCRPS.
Understanding Modern Oncology Trial Designs
The classic, step-by-step path of clinical trials has served us well for decades. But cancer research moves fast, and as we've learned more about the genetics of cancer, trial designs have gotten smarter and more flexible. These newer approaches are all about getting the right drugs to the right patients, faster—a goal that's especially critical when you're facing a complex or treatment-resistant cancer.
Instead of just testing one drug for one cancer type, these modern trials are built differently. They start from the understanding that what truly matters isn't just where a cancer started, but the specific genetic mutation that’s making it grow. This fundamental shift has paved the way for some really innovative trial models.

Basket Trials: One Key for Many Locks
Think of a new drug as a highly specialized key. A traditional trial would test that key on only one type of lock—let's say, melanoma. A basket trial, on the other hand, takes that same key and tests it on many different locks, as long as they all share the same internal mechanism.
In the world of oncology, this means testing a single targeted drug on multiple types of cancer that all happen to share the exact same genetic mutation. For instance, a drug that targets the BRAF V600E mutation could be given to patients with melanoma, colorectal cancer, and thyroid cancer, provided their tumors all carry that specific mutation.
It’s a powerful approach because it focuses on the tumor's molecular fingerprint, not just its address in the body. This "tumor-agnostic" strategy is a game-changer; it’s what led to the first FDA approval of a cancer drug based on a biomarker rather than a cancer type.
Umbrella Trials: One Lock, Many Keys
Now, let's flip that idea. What if you're dealing with one specific type of cancer, but you know it can be caused by a variety of different genetic drivers? That’s where an umbrella trial comes in. Think of it as one very complex lock that might be opened by several different keys.
In an umbrella trial, every participant has the same type of cancer, like non-small cell lung cancer. But first, each person's tumor is genetically sequenced to figure out which mutation is driving their disease. Based on that result, they are assigned to a specific group—a kind of "sub-trial"—that's testing a drug designed to hit their exact mutation.
This setup lets researchers test multiple targeted drugs for a single cancer all at once, under one master plan. It’s an incredibly efficient way to match patients with the treatment that has the best shot at working for them from the get-go.
These master protocols, like basket and umbrella trials, represent a significant shift. They move away from the one-size-fits-all model and toward a future where treatment is defined by a tumor's unique biology, reflecting a core principle of personalized medicine.
Adaptive Trials: A Study That Learns on the Fly
Traditional clinical trials are rigid. The rules are set at the very beginning and rarely change. An adaptive trial, however, is designed to be dynamic. It gives researchers the ability to make pre-planned adjustments to the trial while it's still running, based on the data coming in.
It’s a bit like using a GPS app during a road trip. If the app detects a major traffic jam ahead, it will reroute you in real-time to find a more efficient path to your destination.
Adaptive trials can make several kinds of changes:
- Dropping treatments that aren't working: If a drug in one arm of the trial is proving ineffective, that group can be closed early.
- Expanding successful groups: If another drug is showing real promise, more new patients can be enrolled in that arm to gather more data, faster.
- Adjusting the dosage: Researchers can fine-tune dosages as they learn more about the drug's safety and effectiveness.
This flexibility makes the entire research process faster and more centered on the patient's needs. As patients become more involved in the research process, they are helping to drive this change. You can learn more about how patient advocacy is shaping clinical trial design and pushing for these more responsive models. By learning from results as they happen, adaptive trials help ensure that a patient's time and participation are directed toward the most promising options available.
Asking the Right Questions: What to Discuss With Your Doctor
Considering a clinical trial is a huge step, and it's completely normal to feel a mix of hope and uncertainty. The best way to move forward with confidence is to arm yourself with information. Think of your oncologist and the research team as your partners—and a good partnership starts with a great conversation.
To make sure you get a full picture, it helps to organize your questions. We've broken them down into a few key areas to help guide your discussion, making sure you understand the science, the practicalities, and how the trial fits into your personal care plan.
Understanding the Study's Purpose
First, you want to get a handle on the trial itself. What is it trying to accomplish, and why do the researchers think this new approach might work? This isn't about getting a PhD in molecular biology; it's about understanding the basic goal and the science behind it.
You might ask things like:
- What is the main goal of this study? Are you trying to find a safe dose (Phase I), see if the drug works (Phase II), or compare it to the current standard treatment (Phase III)?
- Why do you believe this treatment could be a better option for me?
- What are the potential benefits you hope to see, and what are the known or suspected risks and side effects?
- How will you actually measure if the treatment is working? Will it be through scans, blood tests, or something else?
Figuring Out the Logistics and Your Commitment
A clinical trial will become a part of your life for a period of time, so it’s essential to understand the practical details. Knowing what to expect day-to-day helps you decide if the schedule and requirements are a good fit for you and your family.
The informed consent form is the start of a conversation, not the end of one. You have the right to ask questions anytime—before, during, or even after you’ve enrolled.
Think about the real-world impact:
- How often will I need to come to the clinic or hospital? Will these be extra visits on top of my usual appointments?
- What specific tests or procedures are involved? Will I need more biopsies, blood draws, or scans than I’m getting now?
- How long is this study expected to last?
- Are any costs covered, like travel, parking, or lodging if I have to travel for treatment?
Focusing on Your Personal Care and Well-Being
Finally, and most importantly, you need to be clear on how the trial will work with your overall cancer care. Your health and well-being are always the #1 priority. You should feel certain that you will be fully supported throughout the process.
Be sure to clarify:
- Can I still see my regular oncologist? How will my care be coordinated between the research team and my doctor?
- Who do I call if I start experiencing side effects? What is the plan for managing them?
- What happens if my cancer gets worse while I’m on the trial?
- If I decide this isn’t for me, can I leave the study at any time? (The answer should always be yes, but it’s a critical right to confirm.)
Having these conversations ensures you’re not just a participant, but an informed partner in your care. To help you keep track, we've put all these key questions into a simple checklist you can bring to your next appointment.
Your Clinical Trial Question Checklist
Use this table to guide your conversation with your doctor and the research team. Don't be afraid to take notes directly on it.
| Category | Question to Ask Your Doctor |
|---|---|
| The Trial's Goal | What is the main purpose of this study (Phase I, II, III)? |
| Why do you think this new treatment might work for me? | |
| What are the biggest potential benefits we're hoping for? | |
| What are all the known risks and possible side effects? | |
| Measuring Success | How will we know if the treatment is effective? |
| What specific tests (scans, bloodwork) will be used to track my progress? | |
| Logistics & Time | How often will I need to be at the clinic? |
| How long is the entire trial expected to last? | |
| Are any of my costs, like travel or parking, reimbursed? | |
| Procedures | What kinds of tests, scans, or procedures are required? |
| Are there more invasive procedures (like biopsies) than in standard care? | |
| Your Healthcare | Will you coordinate with my regular oncologist? |
| Who is my main point of contact for questions or problems? | |
| Side Effects | What is the plan for managing my side effects? |
| Who should I call if I have a problem after hours or on a weekend? | |
| Contingency Plans | What happens if my health declines during the trial? |
| If the treatment doesn't work for me, what are the next steps? | |
| Patient Rights | Can I leave the trial at any time, for any reason? |
| Can I continue my other regular medications while on the trial? |
This list isn't about checking off boxes—it's about opening a dialogue. The goal is for you to walk away from the conversation feeling clear, respected, and ready to make the decision that is truly best for you.
How to Find Cancer Clinical Trials in NYC
When you’re looking for a clinical trial in New York City, the best place to begin is right in your oncologist's office. Your care team knows your medical history inside and out, making them the most qualified resource to connect you with studies that are a good fit, including promising trials right here at Hirschfeld Oncology.
That said, you can and should be your own best advocate. Arming yourself with information by exploring some key online resources will help you have a more productive conversation with your doctor about all the possibilities.

Key Online Resources for Your Search
The go-to database for virtually every registered study in the world is ClinicalTrials.gov. It’s run by the U.S. National Library of Medicine and can feel a bit overwhelming at first. The trick is to use the "Advanced Search" to narrow the results by your specific cancer type, location (like "New York, NY"), and the trial phase you're interested in.
For a more focused search, the National Cancer Institute (NCI) has its own curated list of cancer-specific trials. It’s often a bit easier to navigate and is an excellent way to find NCI-sponsored research happening across NYC's top institutions.
Don’t overlook patient advocacy groups, either. Organizations like the Pancreatic Cancer Action Network (PanCAN) or the Colorectal Cancer Alliance are fantastic. They often have dedicated trial finders tailored to your specific diagnosis and provide a whole ecosystem of support.
The Growing Role of AI in Trial Matching
Finding the right trial is getting easier, thanks to some incredible technology. Artificial intelligence is now being used to sift through vast amounts of medical data and complex tumor biology, making it much faster to pinpoint studies you might be eligible for. It's also making participation easier through remote assessments, which can cut down on travel and time spent at the clinic. You can learn more about the future of clinical trial trends on Merative.
Your Local NYC Advantage: Living in a major research hub like New York City provides direct access to world-class medical centers and a high concentration of clinical trials, especially for novel therapies like immunotherapy.
As you explore these options, you'll likely encounter many different treatment approaches. To get a better handle on one of the most exciting fields in oncology today, check out our guide on finding immunotherapy for cancer near you.
By working closely with your oncologist and using these tools to explore what’s out there, you can take a confident, informed role in planning your next steps.
Frequently Asked Questions About Clinical Trial Phases
Thinking about a clinical trial can bring up a lot of questions. That’s completely normal. Getting straightforward answers is the best way to feel confident and in control of your decisions. Let's walk through some of the most common concerns we hear from patients and their families.
Will I Get a Placebo in a Cancer Trial?
This is probably one of the biggest myths about cancer trials. It is extremely rare for a patient to receive only a placebo, or a "sugar pill." That would only ever happen if there were no existing treatments for a specific cancer.
Instead, a new investigational drug is almost always compared against the current standard of care—the best treatment doctors are already using. Your health is the top priority, and you would never be denied a therapy that is known to be effective.
Can I Leave a Clinical Trial After I Join?
Yes, absolutely. Your participation is 100% voluntary, and you can decide to leave a trial at any point, for any reason.
This will not affect your relationship with your medical team or your future care. If you do decide to stop, your doctor will simply sit down with you to discuss the best alternative treatments available. It’s your right.
At the end of the day, you are always in the driver's seat. The choice to join, continue, or leave a clinical trial is yours and yours alone.
Who Pays for the Costs of a Trial?
The financial side of things can be confusing, but it's usually quite clear. The trial sponsor—typically a pharmaceutical company—covers the cost of the new drug being studied. They also pay for any extra tests, scans, or procedures that are required for the research itself.
Your regular health insurance is generally billed for routine care costs, like the doctor visits and lab work you would be getting anyway as part of standard treatment. A financial coordinator will walk you through everything upfront so there are no surprises.
How Is Patient Safety Protected?
Patient safety isn't just a goal; it's the absolute foundation of every clinical trial. In the United States, every single trial is reviewed, approved, and continuously monitored by an Institutional Review Board (IRB).
Think of the IRB as an independent ethics committee made up of doctors, scientists, and even members of the public. Their entire job is to make sure the trial is designed safely and that any risks are reasonable compared to the potential benefits. On top of that, the dedicated research team will be monitoring your health incredibly closely every step of the way.
At Hirschfeld Oncology, we know that clear, honest conversations are essential to great care. If you have more questions about how clinical trials work or want to explore personalized options for advanced cancer, our team is here to help. You can learn more about our approach at Hirschfeld Oncology's Blog.





.png)


.png)
.png)




