A lot of people first hear the phrase DNA sequencing in a stressful moment. You're sitting in an exam room, trying to keep up, and someone says your tumor may need “sequencing” or “genomic testing.” It can sound technical, expensive, and distant from the immediate question on your mind, which is simple: Will this help me choose the right treatment?
That reaction is completely normal.
When families ask me to define DNA sequencing, they usually aren't asking for a graduate-level science lecture. They want to know what doctors are looking for, why it matters, and whether the results could open doors to better options, especially when standard treatment hasn't worked or has caused too many side effects. That’s the practical question, and it’s the right one.
DNA sequencing matters in cancer care because it can help us read the genetic changes inside a tumor. Some of those changes act like clues. They may show why a cancer is growing, whether a targeted drug could help, or whether an immunotherapy approach makes sense. Not every test finds a clear answer, but when it does, the information can change the direction of care in a meaningful way.
Your Guide to Understanding DNA Sequencing in Cancer Care
A patient may come home from an oncology visit and tell their family, “The doctor wants to send my biopsy for sequencing.” Then the questions start. Is this the same as a biopsy? Does it test my whole body or just the cancer? Does a result mean there’s a treatment, or only more uncertainty?
Those questions deserve plain answers.
DNA sequencing is one of the tools doctors use to understand a cancer more precisely. It doesn't replace your scans, symptoms, pathology report, or physical exam. It adds another layer of information by looking at the tumor’s genetic code and asking whether there are changes that could affect treatment decisions.
Practical rule: You don't need to understand every lab detail. You do need to understand what question the test is trying to answer for your care.
For many patients, the biggest relief comes from realizing this isn't a test you have to “perform well” on. You don't study for it. You don't need to memorize gene names. Your role is usually to provide a sample, ask good questions, and review the results with your care team in a way that connects directly to your treatment choices.
Here’s what often matters most to patients and caregivers:
- What the test reads: It looks at DNA, the body’s genetic instruction code.
- Why doctors order it: To search for changes in the cancer that may guide therapy.
- What results can do: They may support targeted therapy, immunotherapy, or confirm that standard approaches remain the best path.
- What results can't promise: A sequencing test doesn't guarantee a drug match or a cure.
That balance is important. Sequencing can be powerful without being magic. The value is clarity. Sometimes it identifies a treatment opportunity. Sometimes it helps rule one out. Both can prevent wasted time and unnecessary toxicity.
What Is DNA Sequencing in Simple Terms
Think of DNA as a very long instruction book written with just four letters: A, T, C, and G. Every cell in your body carries that book. The order of those letters matters because the sequence tells cells how to function.
DNA sequencing means reading that order.
If you want to define DNA sequencing in everyday language, the simplest answer is this: it’s the process of reading the genetic letters in DNA to see whether the instructions are normal or whether there are meaningful changes. In cancer, those changes can affect how a tumor grows and how it may respond to treatment.

Reading the book one letter at a time
A simple analogy helps. If your body is built from a recipe book, DNA is the full collection of recipes. A gene is one recipe. A mutation is a spelling change in the instructions.
Some spelling changes don't matter. Others do. In cancer, a meaningful change may tell the cell to keep dividing when it should stop. Sequencing helps doctors look for those important errors instead of guessing.
That’s why patients often hear several related terms at once:
- DNA: The genetic material itself.
- Gene: A section of DNA with a specific instruction.
- Mutation or variant: A change in the DNA sequence.
- Sequencing: The act of reading the sequence to find those changes.
If you're also trying to sort out the broader testing environment, this overview of how genetic testing works can help separate inherited testing from other forms of genetic analysis. In cancer care, that distinction matters because some tests look at the tumor itself, while others look for inherited risk.
Why this was such a big scientific breakthrough
For a long time, reading DNA was painstaking work. Earlier methods could sequence only 5 to 10 bases with great difficulty, according to a historical review of sequencing advances in this history of DNA sequencing. The major turning point came when Frederick Sanger developed Sanger sequencing in 1977, a breakthrough that enabled the reading of the first complete genomes and shaped the field for more than 30 years, later paving the way for the Human Genome Project, which took 13 years and cost over $2.7 billion.
That history matters because it shows why sequencing carries so much weight in modern medicine. What once took years and massive resources can now be used in a clinical setting to answer focused questions about a patient’s cancer.
For a deeper look at tumor-focused testing language, this guide on molecular testing for cancer can help clarify how sequencing fits into the larger diagnostic picture.
A useful way to think about sequencing is this: pathology tells us what the cancer looks like, while sequencing can help tell us what the cancer is driven by.
Comparing Major Sequencing Methods Sanger vs NGS
Patients usually don't need to memorize platform names, but it helps to know why one test reads a small region very carefully while another scans much more broadly. In oncology, the two names you'll hear most often are Sanger sequencing and next-generation sequencing, usually shortened to NGS.
The easiest way to separate them is by scale.
One careful proofreader and one large reading team
Sanger sequencing is like one meticulous proofreader reading a short, important passage and checking every letter with extreme care. NGS is like a whole reading team working through many pages at once so the doctor gets a much broader view.
That doesn’t make one “good” and the other “bad.” It means each serves a different purpose.
Sanger is often used when clinicians want to confirm a specific variant in a smaller region. A review from the Federal Judicial Center describes Sanger as the gold-standard first-generation method, with per-base accuracy over 99.999%, read lengths typically 500 to 1000 bp, and cost around $0.50 to $1 per 1000 bp in the cited framework. The same review notes that Sanger remains highly useful for clinical validation of small variants such as EGFR or KRAS, and that its low error rate can outperform short-read NGS in some repetitive regions and heterozygous indels, as explained in this overview of DNA sequencing technologies.
NGS, by contrast, was a major advance because it allowed massive parallel sequencing. Instead of one fragment at a time, it can evaluate many fragments together and build a wider tumor profile.
Sanger Sequencing vs. Next-Generation Sequencing (NGS)
| Feature | Sanger Sequencing ('The Proofreader') | Next-Generation Sequencing ('The Library') |
|---|---|---|
| Main strength | Extremely accurate reading of small, specific regions | Broad analysis of many DNA regions at once |
| Best use in cancer care | Confirming a known or suspected mutation | Profiling a tumor for multiple possible mutations |
| Scale | One targeted stretch at a time | Many fragments in parallel |
| Typical role | Validation and focused follow-up | Discovery and comprehensive testing |
| Read style | Longer reads in a narrow target | High-throughput short-read analysis across many targets |
| Clinical feel | Precise confirmation | Wide search for actionable findings |
Why both methods still matter
If a tumor test suggests a mutation that may guide treatment, doctors sometimes want confirmation before basing a major decision on it. That’s where Sanger still earns trust. The Federal Judicial Center review specifically highlights its role in validating variants before committing to targeted therapy.
NGS becomes especially useful when the question isn't narrow. If a patient has advanced or treatment-resistant cancer, doctors may want to look across many genes at once rather than ordering one small test after another. That broad search can help identify patterns that wouldn't be visible with a single-gene approach.
For readers who want to understand how broad sequencing informs real-world oncology choices, this discussion of next-generation sequencing in clinical decision making for cancer care adds helpful context.
When families ask which method is better, the honest answer is: the better test is the one that matches the clinical question.
The Patient's Journey Through DNA Sequencing
Most patients experience DNA sequencing as a clinical process, not as a laboratory concept. You won't be standing over a machine while DNA is read. What you will notice is a series of decisions, appointments, and follow-up discussions that turn a sample into information your doctor can use.
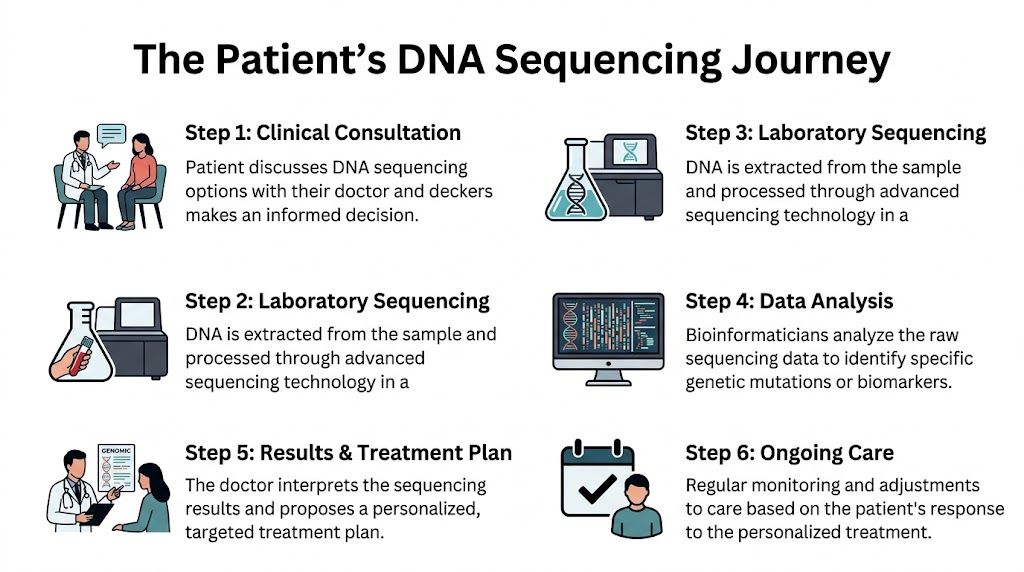
What happens first
The journey usually starts when your oncologist decides that more genetic detail could help guide care. That may happen at diagnosis, after a cancer stops responding to treatment, or when the team is considering targeted therapy or immunotherapy.
At that visit, patients often ask practical questions, not scientific ones. Will this require another procedure? Can my old biopsy be used? Is blood enough, or do I need tissue? Those are exactly the right questions to ask.
What the sample step may look like
The sample may come from tissue already collected during a prior biopsy or surgery. In some cases, doctors may use a blood sample, especially when a liquid biopsy is appropriate.
From the patient’s point of view, this part is usually the most straightforward. After the sample is collected or retrieved, the technical work shifts to the lab.
Here’s the path in plain language:
Clinical decision
Your doctor decides what kind of sequencing question needs answering.Sample collection or retrieval
The team uses stored tumor tissue, fresh biopsy material, or sometimes blood.DNA extraction
Lab specialists isolate the DNA from that sample.Sequencing and analysis
Machines read the genetic material, and bioinformatics experts analyze the data.Clinical interpretation
A report is generated, but the report itself isn't the final answer. Your doctor has to interpret which findings matter.Treatment planning
The care team decides whether the findings support a treatment change, a confirmation step, or no change at all.
Where people often get confused
A sequencing report can list many findings, and not all of them are important. Some are clearly relevant. Some are uncertain. Some may have no treatment implication today.
That’s why the report should never be read as a simple yes-or-no sheet. It needs clinical interpretation. Your doctor looks at the genetics in the context of the whole picture, including the cancer type, stage, prior treatment, symptoms, organ function, and treatment goals.
A sequencing result isn't useful because it is complex. It's useful when a clinician can connect it to a real decision.
What your role is during the process
Patients sometimes feel they should “do more” while waiting. In reality, your main role is to stay engaged and informed.
- Ask what sample is being used: Old tissue and fresh tissue can answer slightly different questions.
- Ask what the team hopes to find: A broad search is different from confirming one known mutation.
- Ask how the result will affect care: That keeps the discussion practical.
- Ask who will explain the report: The key is interpretation, not just receiving paperwork.
The process may feel abstract while the sample is in the lab. Once the results come back, the conversation becomes much more concrete.
How Sequencing Guides Your Cancer Treatment Plan
A sequencing report becomes useful only when it changes what a doctor can do for you. This is its clinical value. We aren't testing for curiosity. We’re looking for information that can help match treatment to the biology of the cancer.

From gene finding to treatment choice
The phrase doctors often use is actionable mutation. That means a genetic change that may point toward a treatment strategy. Sometimes that strategy is a targeted drug. Sometimes it helps identify whether immunotherapy may be more likely to help.
The growth of NGS changed this dramatically. According to Thermo Fisher’s overview of sequencing history, the cost of sequencing a human genome dropped from over $100 million in 2001 to under $1,000 today, and this broader access has helped oncologists identify important biomarkers such as BRCA1/2 mutations in 15 to 20% of breast and ovarian cancers and MSI-high status in about 15% of colorectal cancers, as described in this history of next-generation sequencing.
That drop in cost matters because it moved sequencing from an extraordinary research effort into everyday cancer decision-making.
What this can mean in real cancer care
Different cancers raise different sequencing questions.
In colorectal cancer, an MSI-high result can matter because it may support immunotherapy planning. In breast or ovarian cancer, a BRCA1/2 finding may influence targeted therapy choices. In tumors where a clinician suspects a specific mutation is present, sequencing can help confirm whether that target is present before treatment begins.
Sometimes the most valuable result is a narrow one. It tells the team that a hoped-for target isn't present, which prevents a detour into a treatment unlikely to help. That may sound disappointing, but in practice it can save time, side effects, and false hope.
For families trying to understand how tumor biology affects treatment selection, this guide on how genomic profiling guides therapeutic decisions in oncology offers a useful companion explanation.
Targeted therapy and immunotherapy are different
Patients often hear these terms together, but they aren't the same.
- Targeted therapy aims at a specific molecular feature of the cancer.
- Immunotherapy helps the immune system recognize or attack the tumor more effectively.
- Sequencing can support either path by showing whether the tumor has features linked to response.
A good way to think about it is that sequencing doesn't treat cancer by itself. It improves the map. A better map can lead to a better route.
Here’s a short video that gives additional perspective on how genomic information informs cancer care:
Why this matters for advanced disease
In advanced cancers, treatment decisions often become more individualized over time. A patient may have already received standard therapy. The next question is whether the tumor carries a signal that supports a more personalized approach.
That’s where sequencing can be most helpful. It may identify a reason to consider a targeted option, support the use of immunotherapy, or justify confirming a result before making a treatment change. It also helps doctors avoid assuming that every patient with the same cancer type should receive the same next treatment.
The goal isn't to find more data. The goal is to find the right next step with the least unnecessary toxicity.
Understanding Limitations Accuracy Cost and Privacy
DNA sequencing is powerful, but it has limits. Patients deserve a clear explanation of those limits because trust in testing comes from honesty, not hype.

Accuracy is high, but interpretation still matters
A sequencing platform can be technically strong and still leave room for uncertainty in the final report. The challenge isn't always whether a machine can read DNA. The challenge is whether a detected change is important for that patient’s cancer.
Some parts of the genome are harder to analyze than others. According to a review discussing recent trends, NGS can struggle with certain complex parts of the genome, and newer approaches such as long-read sequencing are being developed to improve diagnostic performance, though access remains uneven, as discussed in this review of long-read sequencing and access issues.
That means a “normal” or “unclear” result doesn't always mean the tumor has no meaningful biology. Sometimes it means today's methods or today's evidence can't fully answer the question.
Cost and insurance can be a real burden
This is often the first practical concern families raise. The same review notes that patients may face $1,000 to over $5,000 for clinical exome sequencing, and that insurance navigation is a major concern in places like New York City. The review also describes sequencing costs as continuing to fall by about 15% annually.
Those trends are encouraging, but they don't erase the stress of coverage decisions.
When cost is part of the conversation, it helps to ask:
- What exact test is being ordered: “Sequencing” can refer to different kinds of testing with different costs.
- Whether prior authorization is needed: Insurance approval may affect timing.
- Whether tissue from a prior biopsy can be used: Reusing a stored sample may reduce additional procedures.
- What the out-of-pocket estimate is: Families should ask for the practical number, not just a general statement.
Privacy questions are reasonable
Genetic information feels personal because it is personal. Patients often worry about who will see the results, how they will be stored, and whether the information could be used beyond direct care.
The right starting point is transparency. Your care team and the lab should explain what kind of test is being ordered, whether it looks only at the tumor or also raises inherited questions, and how the data will be handled. If a result suggests a possible inherited finding, that often leads to a separate conversation about formal germline testing.
If you don't understand what is being tested, ask before the sample is sent. Consent should feel informed, not rushed.
The future is promising, but not equal yet
Long-read sequencing and other newer approaches may improve how we analyze difficult regions of the genome. That's encouraging for the field and for patients with tumors that don't fit neatly into standard testing patterns.
At the same time, access doesn't spread evenly. Some patients can reach advanced genomic testing quickly, while others face delays related to geography, cost, insurance, or referral pathways. So while the technology is advancing, the care system still has work to do.
Accessing Genomic Testing at Hirschfeld Oncology
If you're considering genomic testing, the most useful next step isn't to search for more jargon. It's to bring your own case into focus. The right question is whether sequencing is likely to add something actionable to your treatment decisions now.
At Hirschfeld Oncology, that conversation is especially relevant for patients with advanced, complex, or treatment-resistant cancers. In those settings, sequencing may help clarify whether the tumor has a targetable feature, whether immunotherapy markers are present, or whether a suspected finding should be confirmed before a treatment change.
What that conversation should sound like
A good consultation about sequencing should feel collaborative. You shouldn't leave with only a test name and a consent form. You should leave understanding what the team is looking for and how the result could affect your care.
That includes practical details such as whether stored tissue can be used, whether blood-based testing is appropriate, and what the possible result categories are. It also includes the harder conversation: what the plan will be if no clearly actionable mutation is found.
Here are strong questions to bring to an appointment:
Is my cancer type a good fit for genomic testing right now?
Timing matters. In some cases sequencing is most useful at diagnosis. In others, it becomes more valuable after prior treatment.What do you hope this test will tell us that we don't already know?
This keeps the discussion focused on medical value, not just technology.Will you use prior biopsy tissue, new tissue, or blood?
The sample source can affect what kind of information is available.If the report shows a mutation, how would that change treatment?
This turns a lab concept into a real care decision.If the report doesn't find anything actionable, what comes next?
A good plan includes that possibility from the start.
Why this matters for families too
Caregivers often hear technical terms secondhand and then carry the burden of trying to make sense of them at home. Bringing a written list of questions can help. So can asking for a copy of the report and a plain-language explanation of the findings.
Families don't need to become genetic experts. They do need enough clarity to support decisions about treatment goals, tolerability, and next steps. That matters especially when the patient is weighing options such as targeted therapy, immunotherapy, infusion-based treatment, or a lower-toxicity individualized regimen.
What makes a sequencing discussion worthwhile
A worthwhile discussion does three things at once. It respects the science, it respects the patient’s time, and it respects the patient’s priorities.
That means the conversation should connect the test to symptoms, prior treatment history, treatment goals, and quality of life. For one patient, sequencing may help identify another line of therapy worth pursuing. For another, it may confirm that the best path is symptom control and avoiding burdensome treatment that is unlikely to help.
Both outcomes can be valuable. Precision medicine isn't only about adding treatment. Sometimes it's about avoiding the wrong treatment.
Frequently Asked Questions About DNA Sequencing
Is DNA sequencing the same as genetic testing
Not always. People often use the terms loosely. In cancer care, sequencing may refer to testing the tumor’s DNA to look for treatment-relevant changes. Genetic testing can also refer to inherited testing, which looks for changes present in the DNA you were born with. The distinction matters, and your doctor should explain which type is being ordered.
Does sequencing mean I have a hereditary cancer syndrome
No. A tumor sequencing result does not automatically mean a mutation is inherited. Some changes are found only in the cancer itself. If a result raises concern for an inherited risk, your doctor may recommend separate germline testing and genetic counseling.
Will sequencing definitely find a treatment
No. Some tests identify a clearly actionable target. Others find changes that are interesting but not currently useful. Some don't find a target that changes treatment at all. Even then, the result can still help by narrowing choices and avoiding ineffective options.
Do I need another biopsy
Sometimes yes, sometimes no. Doctors may be able to use tissue already collected from an earlier biopsy or surgery. In other situations, a fresh sample or blood-based testing may be more appropriate. The answer depends on tissue availability, sample quality, and what question the team is trying to answer.
How long does it take to get results
Timing varies by test and lab. The best approach is to ask your care team for the expected turnaround time before the sample is sent. That helps set expectations and can also affect treatment scheduling.
Is a blood test as good as a tissue test
They answer overlapping but not identical questions. A blood-based test can sometimes detect tumor DNA circulating in the bloodstream. Tissue testing may still provide important information that blood testing misses. Doctors choose based on the clinical situation, what tissue is available, and how urgent the treatment decision is.
If I already had sequencing once, can I need it again later
Yes. A cancer can change over time, especially after treatment. A prior result may still be useful, but in some cases doctors repeat testing if the clinical picture changes or if a new treatment question comes up.
What should I bring to an appointment about sequencing
Bring your pathology reports, a treatment history, a list of current medications, and written questions. If you already have a genomic report from another center, bring that too. A second review can be helpful, especially when treatment decisions are complex.
If you or a loved one wants help understanding how genomic testing may fit into a personalized treatment plan, Hirschfeld Oncology offers patient-centered guidance for complex cancer cases, including discussions about targeted therapy, immunotherapy, and lower-toxicity treatment strategies. A thoughtful consultation can help you understand whether sequencing is likely to add useful information for your next decision.





.png)


.png)
.png)




