The first days after a cancer diagnosis often feel unreal. One appointment becomes three. New words show up in your chart. Family members search late into the night, trying to understand what this diagnosis means and what comes next.
For many patients with advanced cancer, the fear isn't only about the disease. It's also about the possibility of being pushed into a treatment plan that feels harsh, generic, and disconnected from the life they still want to live. That concern is valid. Not every treatment fits every tumor, and more treatment is not always better treatment.
The advantages of genetic testing are particularly significant as genetic testing can help doctors look beyond the organ where the cancer started and study the biology driving it. In practical terms, that can open the door to treatments that are more precise, more effective for the right patient, and sometimes less toxic than broad, one-size-fits-all approaches.
Your Cancer Diagnosis Is Not the Whole Story
A diagnosis like pancreatic cancer, bile duct cancer, or metastatic colorectal cancer can sound final when you first hear it. Patients often come in believing the label itself tells us everything we need to know. It doesn't.
Two people can have cancer that starts in the same organ and still have very different disease biology. One tumor may carry a mutation that makes it vulnerable to a targeted drug. Another may have features that make immunotherapy a reasonable option. A third may not show either of those paths, which is also useful to know because it helps avoid chasing treatments that aren't likely to help.

That difference matters emotionally as much as medically. When families understand that a diagnosis is the starting point, not the whole story, the conversation changes. Instead of asking only, “What is the standard treatment?” they begin asking better questions: What is unique about this cancer? What weaknesses does it have? Are there inherited risks we should know about? If you're wondering about the family side of that question, this overview of whether cancer can be genetic is a helpful place to start.
What patients usually need to hear first
During the first discussion, a lecture on molecular biology is unnecessary. Clarity is what's needed.
A useful way to frame it is this:
- The diagnosis names the cancer
- The stage tells us how far it has spread
- Genetic testing can show what may work against it
Genetic testing doesn't guarantee a targeted treatment. It does give us a better chance of choosing treatment based on the cancer's biology instead of guessing from the label alone.
That shift is one of the biggest practical advantages of genetic testing in modern oncology. It turns a broad diagnosis into a more individualized treatment conversation, and for many families, that brings back a sense of direction.
Germline vs Somatic What Your Genes Can Tell Us
When people hear “genetic testing,” they often assume it means one test with one answer. In cancer care, it usually means two different questions.
Germline testing asks whether you were born with an inherited genetic change that may raise cancer risk. Somatic testing asks what genetic changes developed inside the tumor itself over time. Both matter, but they help in different ways.
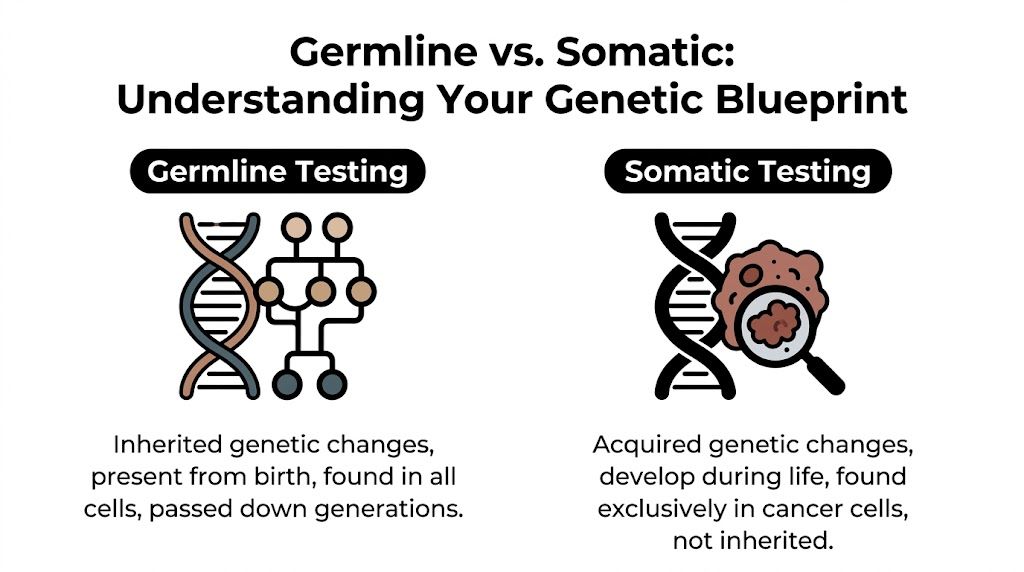
Think blueprint versus renovation
A simple analogy helps.
Your germline DNA is the original blueprint of the house. It's present from birth and found in all your cells. If that blueprint contains an inherited change, it can affect your cancer risk and may also matter to your relatives.
Your somatic tumor DNA is more like damage and renovations that happened later in one room. Those changes were acquired during life, and they're found in the cancer cells, not throughout the body. Those are often the changes that help us match a tumor to a drug.
The differences that matter in practice
| Test type | Usually tested from | Main question | Who benefits most from the result |
|---|---|---|---|
| Germline | Blood or saliva | Did you inherit a cancer-related mutation? | You and your family |
| Somatic | Tumor tissue or sometimes blood-based tumor DNA | What mutations are driving this cancer now? | Your current treatment plan |
This distinction isn't academic. It shapes what we order, what we can learn, and what actions follow.
Why both types can be useful
Advanced testing platforms have improved diagnostic efficiency and, in some cases, eliminated the need for more invasive and costly diagnostic procedures. They also provide actionable information for treatment selection, helping oncologists optimize efficacy while minimizing toxicity, according to this review in PMC on the clinical value of genetic testing.
For patients, that translates into several practical benefits:
- Better treatment matching: Somatic testing may find a mutation that points toward a targeted therapy.
- Family risk clarification: Germline testing can identify hereditary cancer syndromes that affect screening and prevention for relatives.
- Cleaner decision-making: Negative results can matter too. They can help rule out options that sound promising online but don't fit the tumor biology.
- More informed interpretation: If you're curious how raw sequencing data becomes a clinical report, a plain-language explanation of What Is Bioinformatics helps show how labs analyze large sets of genetic information.
Practical rule: Germline results tell us what risk may run in the family. Somatic results tell us what the tumor may respond to right now.
Patients sometimes worry that more testing means more confusion. Sometimes it does create nuance. But most of the time, it replaces vague decision-making with sharper choices.
Unlocking Precision Treatments with Genetic Insights
The strongest clinical argument for testing in advanced cancer is simple. Sometimes a mutation reveals a treatment that fits the tumor far better than standard therapy.
That's where the most important advantages of genetic testing show up. The goal isn't to collect interesting data. The goal is to find an option that gives a patient a better balance of effectiveness and tolerability.

How the lock and key model works
Many cancers grow because a particular pathway is switched on or a repair system is broken. If testing identifies that weakness, treatment can be chosen to exploit it.
A useful metaphor is lock and key. The mutation is the lock. The drug is the key. If you don't know the lock is there, you're left trying treatments that may be broader, harsher, or less effective for that specific cancer.
That doesn't mean targeted therapy is always gentle or always superior. Every drug has side effects. Some patients still need chemotherapy, combinations, or treatment sequences that change over time. But when a well-matched target exists, the treatment plan often becomes more rational.
Pancreatic cancer shows why this matters
In pancreatic cancer, germline testing identifies inherited pathogenic variants in about 10% of cases, and that information can open the door to precision treatment. For patients with BRCA1/2 mutations, PARP inhibitors such as olaparib have been shown to significantly prolong progression-free survival, which is one reason guidance supports testing for all pancreatic cancer diagnoses regardless of family history, as explained in Michigan Medicine's review of why every pancreatic cancer patient should consider genetic testing.
This is more than a lab result. It's a treatment consequence.
When BRCA-related DNA repair is impaired, the tumor may become especially vulnerable to PARP inhibition. That can create a path toward a treatment that is biologically matched to the cancer rather than selected only because it's part of a standard sequence.
For patients and families, this changes the discussion in a very practical way:
- It can identify a targeted option when the cancer has a repair defect.
- It can support treatment choices with a different side effect profile than traditional cytotoxic therapy.
- It can justify testing even without family history, which matters because inherited risk isn't always obvious from a family tree.
If you want a fuller explanation of how tumor sequencing guides these decisions, this overview of molecular testing for cancer is a useful companion.
Other advanced cancers can also reveal actionable targets
Pancreatic cancer isn't the only example. In advanced gastrointestinal cancers, panel testing can uncover alterations that redirect treatment in meaningful ways.
A review of genomic testing in gastrointestinal cancers reports that multigene panel testing reveals actionable somatic and germline alterations in up to 20% to 30% of advanced cases. In metastatic colorectal cancer with BRAF V600E mutations, a testing-guided regimen using encorafenib plus cetuximab and binimetinib achieved median progression-free survival of 9.3 months versus 5.6 months with standard irinotecan-based regimens. The same review notes that identifying NTRK fusions in cholangiocarcinoma or gastric cancers can lead to larotrectinib, with objective response rates of 75% and median progression-free survival of 28 months. It also describes MSI-high disease as a biomarker for response to pembrolizumab in certain settings, all in this PMC review on multigene panel testing in gastrointestinal cancers.
Those details matter because they show what “precision oncology” means. It isn't a slogan. It means a report can change the drug list.
A short explainer can help put this into context:
What works and what doesn't
The best use of genetic testing is disciplined, not wishful.
What tends to work:
- Testing early enough that results can influence treatment choices
- Using broad enough panels when the cancer type justifies it
- Matching results to real treatment options, not just interesting findings
- Reviewing results in context of prior therapies, symptoms, and patient goals
What doesn't work:
- Treating every mutation as actionable
- Assuming targeted therapy has no side effects
- Ordering testing without a plan for how the result will be used
- Chasing a weak signal when the patient's condition calls for a faster, simpler decision
The right test can narrow the field. It doesn't replace judgment. It sharpens it.
That distinction is important. Precision treatment isn't about doing more. It's about avoiding unnecessary treatment and choosing the treatment that fits the tumor in front of you.
More Than Medicine The Broader Benefits of Testing
Treatment matching gets most of the attention, but that's not the only reason testing matters. A genetic result can change how a family understands risk, how future screening is planned, and how a patient thinks about the meaning of this diagnosis.
For some families, one of the hardest parts of cancer is uncertainty. Not just “What medicine comes next?” but “Could this affect my children, my siblings, or my parents?” Germline testing doesn't answer every question, but it can turn vague worry into something specific enough to act on.

Family protection can begin with one result
A landmark Mayo Clinic study found that over 10% of people who underwent predictive genomic testing discovered a hereditary risk for a health condition. About half had no personal or family medical history suggesting that risk, meaning it likely would have gone undetected through conventional methods. The study also found that interest in testing increased from 11% in 2014 to 40% in 2019 among patients at Mayo's Predictive Genomics Clinic, as described in Mayo Clinic's discussion of the benefits of genetic testing for healthy people.
That matters in cancer care because family history isn't always a reliable filter. Some inherited risk is silent until someone in the family gets sick. A positive germline result can allow relatives to pursue their own testing and screening rather than waiting for cancer to announce itself.
The benefit isn't only inherited risk
The broader value of testing often falls into a few real-world categories:
- Eligibility for clinical trials: Some studies require a specific mutation or biomarker. A test result can create access that wouldn't otherwise exist.
- Clearer planning: Certain findings may help frame prognosis or influence how aggressively to pursue one strategy over another.
- Family cascade testing: When one person is found to carry a hereditary mutation, relatives can decide whether to be tested for that same mutation.
- Earlier surveillance: A relative who learns they carry a hereditary risk may begin earlier or more focused screening.
A genetic result can protect more than one person. That's one of the most meaningful parts of this work.
What families should do with the information
A good result review doesn't end with the patient. It usually leads to a conversation like this:
- Clarify whether the finding is inherited or tumor-only
- Identify which relatives may be affected
- Recommend genetic counseling when needed
- Translate the result into screening or prevention steps
Some patients hesitate because they don't want to burden their family. In practice, most relatives would rather know than remain in the dark. Knowledge doesn't remove fear completely, but it often replaces helplessness with options.
A Realistic Look at Genetic Testing's Limitations
Genetic testing is powerful, but it isn't a magic answer. Some patients expect a result that points neatly to one perfect drug. Sometimes that happens. Often it doesn't.
A common frustration is the variant of uncertain significance, often called a VUS. That means the lab found a genetic change, but current science can't say with confidence whether that change is harmful, harmless, or clinically meaningful. Patients understandably hear “something abnormal was found” and assume action is needed. Often, it isn't.
Where uncertainty shows up
A result may be limited for several reasons:
- The finding isn't actionable: A mutation may be real but not linked to an available treatment.
- The evidence is incomplete: Some alterations are still being studied and don't yet justify a treatment decision.
- The sample has limits: Tumor tissue can be small, old, or technically difficult to analyze.
- The biology is complex: Cancers rarely depend on only one mutation. A useful target can still be overridden by resistance pathways.
That doesn't make the test a mistake. It means interpretation matters as much as the sequencing itself.
The emotional side is real
Learning about hereditary risk can stir up guilt, fear, or anger. Parents may worry about children. Siblings may feel pressured to get tested. Some people don't want to know. Others want every detail immediately.
Not every patient needs the same amount of information on the same day. Good care respects both the science and the emotional timing.
Privacy concerns also come up often. Patients should know that GINA, the Genetic Information Nondiscrimination Act, provides important protections in health insurance and employment. It does not erase every concern people may have, and questions about privacy and broader insurance implications deserve careful discussion before testing.
The honest view is this: genetic testing helps most when it's paired with clear counseling, realistic expectations, and careful interpretation. The test alone isn't the solution. The quality of the conversation around it is what makes it useful.
How We Integrate Genetic Testing into Your Care
Good oncology care doesn't treat genetic testing like a box to check. The question isn't whether more data can be ordered. The question is whether the right test, at the right time, will improve treatment selection and quality of life.
That matters most in complex cancers where the next decision carries real trade-offs. A broad panel may be appropriate in one patient with advanced gastrointestinal cancer. In another patient, a more focused approach may be better because the immediate need is speed, symptom control, or confirmation of one specific biomarker.
We choose the test based on the decision in front of us
When testing is integrated thoughtfully, the process usually includes:
- Cancer-specific selection: Pancreatic, colorectal, cholangiocarcinoma, gastric, breast, and ovarian cancers don't all need the same testing strategy.
- Treatment-history review: Prior chemotherapy, immunotherapy, surgery, and progression patterns affect which result will matter now.
- Specimen planning: Sometimes archived tumor tissue is enough. Sometimes a new biopsy or blood-based testing is more practical.
- Action planning before ordering: The result should answer a treatment question, not just satisfy curiosity.
This approach is especially relevant in advanced gastrointestinal cancers. Multigene panel testing reveals actionable alterations in up to 20% to 30% of cases. In metastatic colorectal cancer with BRAF V600E mutations, testing-guided triplet therapy can nearly double progression-free survival compared with standard care. Identifying NTRK fusions in cholangiocarcinoma can lead to therapies with response rates over 75%, supporting a move toward genomic-driven, lower-toxicity treatment selection, as described in the earlier cited PMC review of multigene testing in gastrointestinal cancers.
Results have to be translated, not just delivered
A lab report by itself can be confusing even for medically knowledgeable families. What matters is interpretation.
A useful review of results should answer practical questions such as:
| Question | Why it matters |
|---|---|
| Is this inherited or only in the tumor? | It changes the implications for family members |
| Does this change treatment now? | Some findings are immediately actionable, others are not |
| What side effects should we weigh? | Precision medicine still involves trade-offs |
| What if the result is negative or unclear? | That may still narrow choices and avoid weak options |
Experience is paramount. The goal isn't to impress patients with molecular terminology. The goal is to convert a technical report into a plan that fits the person's disease, symptoms, goals, and tolerance for treatment.
Your Genetic Testing Questions Answered
Will insurance cover genetic testing
Coverage depends on the type of test, the cancer diagnosis, and the clinical reason for ordering it. Many patients are eligible for coverage when testing is medically indicated, but the details vary. Before ordering, ask what the test is meant to answer and whether prior authorization or financial review is needed.
Is the test difficult
Usually, no. Germline testing is often done with a blood draw or saliva sample. Somatic testing commonly uses tumor tissue that has already been collected, although sometimes another biopsy or a blood-based tumor DNA test is considered.
How long do results take
Timing varies by lab and by test type. In practice, the important question isn't only when the report arrives. It's when someone knowledgeable will sit down with you and explain what the result changes, what it doesn't change, and whether your family needs follow-up.
What if my result is unclear
That can happen. A result may show no actionable mutation, or it may find a variant whose significance isn't fully understood. That doesn't mean the process failed. It still helps refine the decision-making and can prevent treatment choices based on assumptions.
How do I make sense of the report
Don't try to decode it alone. Ask for a clear explanation in plain language. This guide to understanding genetic test results can help you prepare better questions for that conversation.
Is my privacy protected
Patients have important protections under GINA for health insurance and employment. Even so, privacy, documentation, and family implications are worth discussing before testing so you understand the practical considerations and can make an informed decision.
If you're weighing treatment options and want a clearer sense of whether genetic testing could open the door to a more precise, less toxic plan, Hirschfeld Oncology offers thoughtful guidance for patients and families facing complex cancers. A focused consultation can help you understand which tests matter, what the results could change, and how to align every next step with both medical goals and quality of life.





.png)


.png)
.png)




